Amity University Noida-B.Tech Admissions 2026
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
JEE Advanced is one of the most competitive exams in India, which is prepared to test knowledge and problem-solving skills. In the JEE Advanced exam, some questions were very difficult and lengthy to solve. This article covers the JEE Advanced most unattempted questions across the physics, chemistry, and maths sections. These unattempted questions help students to determine what type of questions can be considered as JEE Advanced 2026 most unattempted questions. By analysing these toughest questions of JEE Advanced maths, physics, and chemistry, students gain a deeper understanding of the difficulty level and trends that IITs can follow in framing JEE Advanced Paper 2026. Scroll down to know more about the JEE Advanced question paper with solutions PDF, JEE Advanced practice questions chapter-wise, and the toughest questions of JEE Advanced.
This Story also Contains

Some JEE Advanced Chemistry questions were left unanswered. JEE Advanced 2025 Chemistry Analysis helps identify the toughest section of JEE Advanced Chemistry and topics that are difficult to understand
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q13 |
40.49 |
Aldehydes, Ketones, And Carboxylic Acid |
Williamson's ether synthesis |
|
Q10 |
37.97 |
States of Matter |
Vander Waals equation |
|
Q 14 |
35.91 |
P- Block Elements |
Salt analysis |
|
Q 16 |
35.23 |
Amines |
Tests for functional groups |
Question: The reaction sequence given below is carried out with 16 moles of $\mathbf{X}$. The yield of the major product in each step is given below the product in parentheses. The amount (in grams) of $\mathbf{S}$ produced is $\_\_\_\_$ .
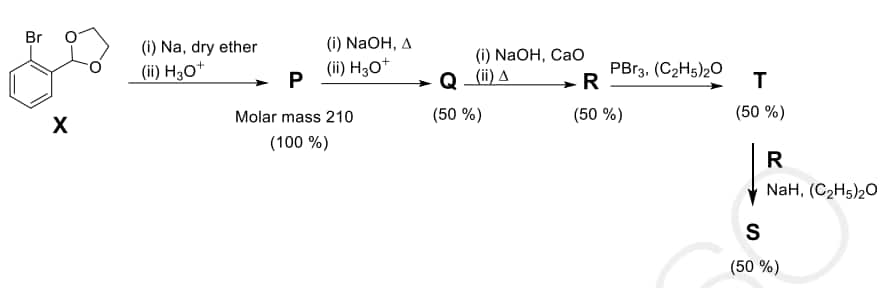
Use: Atomic mass (in amu): $\mathrm{H}=1, \mathrm{C}=12, \mathrm{O}=16, \mathrm{Br}=80$
Solution:
X $\rightarrow$ P:
(i) Na, dry ether:
Molar mass = 210 means two units of the benzene ring must be coupling. Sodium in ether with an aryl bromide leads to the formation of a biphenyl derivative.
(ii) $H_3O^+$:
Structure P: 2,2'-biphenyldicarbaldehyde.
Check Molar Mass: $C_{14}H_{10}O_2 = (14 \times 12) + (10 \times 1) + (2 \times 16) = 168 + 10 + 32 = 210$ g/mol. (Matches).
P $\rightarrow$ Q:
NaOH, $\Delta$ followed by $H_3O^+$: This is an Intramolecular Cannizzaro Reaction NaOH and heat on a dialdehyde like this, it undergoes a transformation to a hydroxy-acid or cyclic structure. $Q$ is likely the product of a cyclization or specific redox shift. $Q$ is 9-fluorenone or a related carboxylic acid.
NaOH/heat followed by decarboxylation (Step R) suggests $Q$ is Biphenyl-2,2'-dicarboxylic acid.
Q $\rightarrow$ R:
NaOH, CaO, $\Delta$: Soda-lime Decarboxylation.
Structure R: Biphenyl
$P$ is the dialdehyde. $Q$ is the result of an internal redox/condensation. $R$ is Fluoren-9-ol.
R $\rightarrow$ T:
$PBr_3$: Converts the alcohol to a bromide.
Structure T: 9-bromofluorene.
T + R $\rightarrow$ S:
NaH, ether: NaH acts as a base to deprotonate the alcohol $R$ (Fluoren-9-ol) to form an alkoxide. This then performs a nucleophilic substitution ($S_N2$) on the bromide $T$.
Structure S: An ether linkage between two fluorene moieties (9,9'-oxybis(9H-fluorene)).
Formula of S: Two fluorene units ($C_{13}H_8 \times 2$) linked by an oxygen. $C_{26}H_{18}O$.
Molar Mass of S: $(26 \times 12) + (18 \times 1) + 16 = 312 + 18 + 16 = 346$ g/mol.
Moles of X =16 moles
Moles of P: $16 \text{ moles of } X \times \frac{1 \text{ unit } P}{2 \text{ units } X} \times 100\% = \mathbf{8 \text{ moles of } P}$
Moles of Q: $8 \text{ moles} \times 50\% = \mathbf{4 \text{ moles of } Q}$
Moles of R: $4 \text{ moles} \times 50\% = \mathbf{2 \text{ moles of } R}$
Moles of T: $2 \text{ moles (available R)} \times 50\% \text{ yield} = \mathbf{1 \text{ mole of } T}$
(R + T $\rightarrow$ S):
We have 1 mole of T remaining and 1 mole of R remaining
The reaction is $R + T \rightarrow S$. This is a 1:1 stoichiometry.
Theoretical yield of S: 1 mole.
Actual yield of S (50%): $1 \text{ mole} \times 50\% = \mathbf{0.5 \text{ moles of } S}$.
Molar Mass of S ($C_{26}H_{18}O$): 346 g/mol
Moles of S: 0.5 mol
Mass of S: $0.5 \text{ mol} \times 346 \text{ g/mol} = \mathbf{173 \text{ grams}}$
The amount of S produced is 173 grams.
Hence, the answer is 173 gm.
Based on the analysis of JEE Advanced and the specific structure of this problem, the following reasons are given why Question 13 saw such a high not-attempted rate.
1. This question uses sequential reactions where the product of one step is the reactant for the next.
2. This question combines Cannizzaro or Wurtz-Fittig reactions in an intramolecular context:
The Cannizzaro Step (P → Q)
Decarboxylation (Q → R)
3. Concepts like stoichiometric ratios, compounding yields, and limiting reagents are used in this question.
4. The ($R + T \rightarrow S$) is a Williamson ether synthesis.
5. This question covers multiple steps and reactions. Those are difficult and time-consuming questions
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q7 |
44.27 |
Amines |
Gabriel phthalamide synthesis |
|
Q5 |
42.53 |
States of Matter |
Intermolecular forces |
|
Q11 |
35.61 |
Surface Chemistry |
Freundlich adsorption isotherm |
|
Q3 |
34.59 |
Aldehydes, Ketones And Carboxylic Acid |
Named reactions Aldol condensation, oxymercuration, ozonolysis |
Question 7 of Chemistry JEE Advanced is one of the key problems that tests both conceptual clarity and application skills

Solution:
Oxidation
Tetralin on strong oxidation with acidic permanganate forms phthalic acid, which on heating with ammonia forms phthalimide.
$\text{Tetralin} \xrightarrow[ \Delta]{KMnO_4/H^+} C_6H_4(COOH)_2$
$C_6H_4(COOH)_2 + NH_3 \xrightarrow{\Delta} C_6H_4(CO)_2NH + 2H_2O$
Thus,
$X = \text{Phthalimide}$
Gabriel synthesis
Deprotonation and alkylation:
$C_6H_4(CO)_2NH + KOH \rightarrow C_6H_4(CO)_2N^-K^+ + H_2O$
$C_6H_4(CO)_2N^-K^+ + RBr \rightarrow C_6H_4(CO)_2NR$
$Y = \text{N-alkyl phthalimide}$
Hydrolysis
$C_6H_4(CO)_2NR + NaOH \rightarrow C_6H_4(COONa)_2 + RNH_2$
$Z = RNH_2$
(A) $X$ and $Y$ contain carbonyl groups → True
(B) Carbylamine reaction:
$
RNH_2 + CHCl_3 + 3KOH \rightarrow RNC + 3KCl + 3H_2O
$
Occurs only for primary amines, not $Y$ → False
(C) $Z = RNH_2$ reacts with Hinsberg reagent → True
(D) $Z$ is aromatic primary amine → False
$\boxed{A \text{ and } C}$
Hence, the correct answers are option 1,3.
1. The first step to solve this question is Multi step Conversion where the reaction requires linking oxidation → phthalimide formation → Gabriel synthesis → hydrolysis.
2. The key intermediate here is phthalimide, which is not a very common compound in everyday practice questions. Since many students had not memorized that naphthalene oxidation → phthalic acid → phthalimide, they struggled to identify “X” confidently.
3. In this question Option (B) tempted students with the carbylamine test, but the intermediate Y is not a primary amine. While option (D) suggested Z is an aromatic amine, but Gabriel synthesis doesn’t yield aryl amines.
4. This question was placed among organic reaction sequence questions, which are usually time-consuming. Many aspirants skipped it to avoid losing time on an unfamiliar, lengthy pathway, preferring quicker physical or inorganic chemistry questions.
More Details check the video
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
The most skipped questions in JEE Advanced Maths were primarily from topics like Matrices, 3D Geometry, Probability, and Integral Calculus. For example, in Paper 1, Q4 (Matrices) had a 72.11% skip rate, while in Paper 2, Q6 (Integral Calculus) was skipped by 78.55% of candidates. For more information refer to JEE Advanced 2025 Mathematics Analysis.
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q4 |
72.11 |
Matrices and determinants |
Transpose of a matrix |
|
Q5 |
62.22 |
Three-dimensional geometry |
Line of Intersection of Two Planes and Angle Between a Line and a Plane |
|
Q1 |
62.1 |
Binomial Theorem and Its Simple Applications |
General Term of Binomial Expansion |
|
Q6 |
55.09 |
Sets, Relations and Functions |
Onto Function or Surjective |
Question: Consider the matrix
$P=\left(\begin{array}{lll}2 & 0 & 0 \\ 0 & 2 & 0 \\ 0 & 0 & 3\end{array}\right)$
Let the transpose of a matrix $X$ be denoted by $X^T$. Then the number of $3 \times 3$ invertible matrices $Q$ with integer entries, such that
$Q^{-1}=Q^T \text { and } P Q=Q P,$
(1) 32
(2) 8
(3) 16
(4) 24
Solution: $Q^{-1} = Q^T$, which implies $Q Q^T = I$ and $Q^T Q = I$. Means $Q$ is an orthogonal matrix.
$PQ = QP$. Since $P$ is a diagonal matrix.
Commutativity ($PQ = QP$)
The matrix $P$ is given as:
$P = \begin{pmatrix} 2 & 0 & 0 \\ 0 & 2 & 0 \\ 0 & 0 & 3 \end{pmatrix}$
Let $Q = \begin{pmatrix} q_{11} & q_{12} & q_{13} \\ q_{21} & q_{22} & q_{23} \\ q_{31} & q_{32} & q_{33} \end{pmatrix}$. The condition $PQ = QP$ implies:
$\begin{pmatrix} 2q_{11} & 2q_{12} & 2q_{13} \\ 2q_{21} & 2q_{22} & 2q_{23} \\ 3q_{31} & 3q_{32} & 3q_{33} \end{pmatrix} = \begin{pmatrix} 2q_{11} & 2q_{12} & 3q_{13} \\ 2q_{21} & 2q_{22} & 3q_{23} \\ 2q_{31} & 2q_{32} & 3q_{33} \end{pmatrix}$
$2q_{13} = 3q_{13} \implies q_{13} = 0$
$2q_{23} = 3q_{23} \implies q_{23} = 0$
$3q_{31} = 2q_{31} \implies q_{31} = 0$
$3q_{32} = 2q_{32} \implies q_{32} = 0$
$Q = \begin{pmatrix} A & 0 \\ 0 & d \end{pmatrix} = \begin{pmatrix} q_{11} & q_{12} & 0 \\ q_{21} & q_{22} & 0 \\ 0 & 0 & q_{33} \end{pmatrix}$
where $A = \begin{pmatrix} q_{11} & q_{12} \\ q_{21} & q_{22} \end{pmatrix}$ is a $2 \times 2$ sub-matrix.
$q_{33}$:
The third column is $(0, 0, q_{33})^T$. For its magnitude to be $1$:
$0^2 + 0^2 + q_{33}^2 = 1 \implies q_{33}^2 = 1 \implies q_{33} = \pm 1$
This gives 2 choices for $q_{33}$.
For the sub-matrix $A$:
$A$ must be a $2 \times 2$ orthogonal matrix with integer entries:
$A^T A = \begin{pmatrix} 1 & 0 \\ 0 & 1 \end{pmatrix}$
$\begin{pmatrix} 1 \\ 0 \end{pmatrix}, \begin{pmatrix} -1 \\ 0 \end{pmatrix}, \begin{pmatrix} 0 \\ 1 \end{pmatrix}, \begin{pmatrix} 0 \\ -1 \end{pmatrix}$
Because $A$ must be orthogonal, the second column must be orthogonal to the first.
This results in the following 8 possible matrices for $A$:
$\begin{pmatrix} 1 & 0 \\ 0 & 1 \end{pmatrix}, \begin{pmatrix} 1 & 0 \\ 0 & -1 \end{pmatrix}, \begin{pmatrix} -1 & 0 \\ 0 & 1 \end{pmatrix}, \begin{pmatrix} -1 & 0 \\ 0 & -1 \end{pmatrix}, \begin{pmatrix} 0 & 1 \\ 1 & 0 \end{pmatrix}, \begin{pmatrix} 0 & 1 \\ -1 & 0 \end{pmatrix}, \begin{pmatrix} 0 & -1 \\ 1 & 0 \end{pmatrix}, \begin{pmatrix} 0 & -1 \\ -1 & 0 \end{pmatrix}$
This gives 8 choices for $A$.
Total number of matrices $Q$ is
$\text{Total } Q = (\text{Choices for } A) \times (\text{Choices for } q_{33})$
$\text{Total } Q = 8 \times 2 = 16$
There are 16 such $3 \times 3$ invertible matrices $Q$.
Hence, the answer is 16.
Refer to the points given below on why students skipped this question:
1. Due to confusion whether matrix multiplication is commutative or not ($PQ \neq QP$).
2. $Q^{-1} = Q^T$ this is the condition of Orthogonal Matrix. Instead of identifying this students try to manually find the inverse ($Q^{-1}$) using the adjoint method. That process becomes lengthy.
3. An orthogonal matrix involves square roots. The requirement for integer entries forces the entries to be only 0, 1, or -1.
4. The process of counting the possible $2 \times 2$ matrices ($A$) leads to lots of errors.
Also Check:
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q6 |
78.55 |
Integral calculus |
Area Bounded by Two Curves |
|
Q3 |
67.16 |
Trigonometry |
Inverse Trigonometric Function |
|
Q7 |
66.7 |
Coordinate geometry |
Line and the Ellipse |
|
Q5 |
63.49 |
Matrices and determinants |
Properties of the Determinant of a Matrix |
In Paper I, questions with the highest percentage of skipped responses were Q4 , Q5 and Q1 . The least skipped response was for Q2. Paper 2, on the other hand, had even higher skips, with Q6 being the most skipped question, touching 141,728 candidates, followed by Q3 and Q7. From this table it is clear that Paper 2 questions had a higher percentage of non attempts compared to Paper.
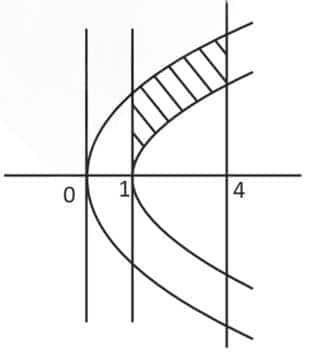
Question: Let $S$ denote the locus of the mid-points of those chords of the parabola $y^2=x$, such that the area of the region enclosed between the parabola and the chord is $\frac{4}{3}$. Let $R$ denote the region lying in the first quadrant, enclosed by the parabola $y^2=x$, the curve $S$, and the lines $x=1$ and $x=4$.
Then which of the following statements is (are) TRUE?
$1-(4, \sqrt{3}) \in S$ (Correct)
$2-(5, \sqrt{2}) \in S$
3- Area of $R$ is $\frac{14}{3}-2 \sqrt{3}$ (Correct)
4- Area of $R$ is $\frac{14}{3}-\sqrt{3}$
Solution:-
Given: The parabola is $y^2=x$
Let a chord of this parabola have midpoint $(h, k)$. The equation of the chord with given midpoint in terms of $T=S_1$ form is:
$
\begin{aligned}
& y k-\frac{y+h}{2}=k^2-h \\
& 2 k y-y^2-h=2 k^2-2 h \\
& \text { Now, } A=\int_{y_1}^{y_2}\left(2 k y-2 k^2+h 0-y^2\right) d y=\frac{4}{3} \\
& \left(k y^2+\left(h-2 k^2\right) y-\frac{y^3}{3}\right)_{y_1}^{y_2}=\frac{4}{3} \\
& \left(y_2-y_1\right)\left[k-2 k+h-2 k^2-\frac{1}{3}\left(4 k^2-2 k^2+h\right)\right]=\frac{4}{3} \\
& \left(h-k^2\right)^{\frac{3}{2}}=1 \\
& h-k^2=1
\end{aligned}
$
Thus, the locus $S$ is:
$x-y^2=1 \Rightarrow y^2=x-1$
We'll check if these points lie on the line, by putting the values,
$(\sqrt{3})^2=4-1 \text {---True }$
But $5^2=\sqrt{2}-1$----False,
Thus, point $(4, \sqrt{3})$ lies on the line, so Option 1 is correct.
Now find the area $R$ enclosed between:
- Parabola $y^2=x \Rightarrow y=\sqrt{x}$
- Curve $S: y=\sqrt{x-1}$
- Vertical lines $x=1$ and $x=4$
The area of region $R$ is:
$\begin{aligned}
& A=\int_1^4(\sqrt{x}-\sqrt{x-1}) d x \\
& =\left[\frac{2}{3} x^{3 / 2}-\frac{2}{3}(x-1)^{3 / 2}\right]_1^4=\frac{2}{3}[8-3 \sqrt{3}-1]=\frac{2}{3}(7-3 \sqrt{3}) \\
& R=\frac{14}{3}-2 \sqrt{3}
\end{aligned}$
Option 3 is also correct
Hence, the correct answers are option 1,3.
Double Concept
In this question two ideas are mixed using parameters for the parabola and then solving with definite integration.
Confusion with Region in First Quadrant
When students read this part, they have to imagine the shapes of the curves $y^2=x$ and $x=y^2+1$ on a graph. Many get confused about which curve lies above or below the other.
Two-Stage Question Format
Even after finding the locus S, the problem is not finished. It also asks to check which points lie on S.
Multiple Correct Answer-Type
The question has options (A–D) where more than one can be correct. With so many steps: locus, integration, and checking points, the chances of making a mistake are high, so many skip it.
Indirect Parametrisation Trick
The simple method is to use parameters for the parabola: $x=p^2, y=p$.
The question looks too lengthy, indirect and too integration-heavy at first sight. It involves two different conceptual tricks like parametrisation of a parabola, simplification of the area formula.
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q1 |
72.08 |
Rotational Motion |
Small Oscillations of a Rolling Body |
|
Q7 |
65.52 |
Oscillations and Waves |
Reflection and Transmission of Waves at a Boundary |
|
Q5 |
60.08 |
Electromagnetic Induction and Alternating Currents |
Eddy Currents |
|
Q2 |
52.53 |
Rotational Motion |
2D Elastic Collision |
Question: The center of a disk of radius $r$ and mass $m$ is attached to a spring of spring constant $k$, inside a ring of radius $R>r$ as shown in the figure. The other end of the spring is attached on the periphery of the ring. Both the ring and the disk are in the same vertical plane. The disk can only roll along the inside periphery of the ring, without slipping. The spring can only be stretched or compressed along the periphery of the ring, following Hooke's law. In equilibrium, the disk is at the bottom of the ring. Assuming small displacement of the disc, the time period of oscillation of center of mass of the disk is written as $T=\frac{2 \pi}{\omega}$. The correct expression for $\omega$ is ( $g$ is the acceleration due to gravity):
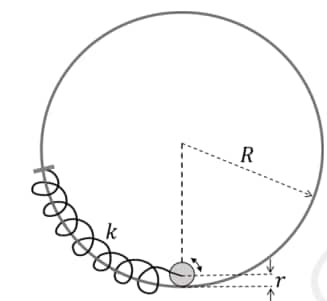
(1) $\sqrt{\frac{2}{3}\left(\frac{g}{R-r}+\frac{k}{m}\right)}$
(2) $\sqrt{\frac{2 g}{3(R-r)}+\frac{k}{m}}$
(3) $\sqrt{\frac{1}{6}\left(\frac{g}{R-r}+\frac{k}{m}\right)}$
(4) $\sqrt{\frac{1}{4}\left(\frac{g}{R-r}+\frac{k}{m}\right)}$
Solution:
Velocity of CM: $v = (R - r)\dot{\theta}$
Angular velocity of disk: $\omega_{d} = \frac{v}{r} = \frac{(R - r)\dot{\theta}}{r}$
$K = \frac{1}{2}mv^2 + \frac{1}{2}I\omega_{d}^2$
Using $I = \frac{1}{2}mr^2$:
$K = \frac{1}{2}m(R - r)^2\dot{\theta}^2 + \frac{1}{4}m(R - r)^2\dot{\theta}^2 = \frac{3}{4}m(R - r)^2\dot{\theta}^2$
For small $\theta$:
Gravity: $U_g \approx \frac{1}{2}mg(R - r)\theta^2$
Spring: $U_s \approx \frac{1}{2}k[(R - r)\theta]^2$ (Spring acts on the CM)
Total Energy $E = K + U$. Setting $\frac{dE}{dt} = 0$:
$\frac{3}{2}m(R - r)^2\ddot{\theta} + [mg(R - r) + k(R - r)^2]\theta = 0$
$\ddot{\theta} + \frac{2}{3} \left[ \frac{g}{R - r} + \frac{k}{m} \right] \theta = 0$
Comparing with $\ddot{\theta} + \omega^2\theta = 0$:
$\omega = \sqrt{\frac{2}{3} \left( \frac{g}{R - r} + \frac{k}{m} \right)}$
Hence, the correct answer is option (1).
Below are reasons students did not attempt this question:
1. This question is related to the Rolling without Slipping condition on a curved surface.Many students incorrectly assume the angular velocity of the disk.
2. Spring is attached to the center of the disk but follows the periphery of the ring. Students get confused about whether the extension is $R\theta$ or $(R-r)\theta$.
3. Sum of three different types of energy is to be calculated to solve this question:
Translational Kinetic Energy: $\frac{1}{2}mv^2$
Rotational Kinetic Energy: $\frac{1}{2}I\omega^2$
Potential Energy: Both gravitational ($mgh$) and elastic ($\frac{1}{2}kx^2$)
Recognized as Institute of Eminence by Govt. of India | NAAC ‘A++’ Grade | Upto 75% Scholarships | Only JEE Scores Accepted
Ranked #29 in Engineering Category in India by NIRF Ranking 2025 | Highest CTC 1.23 Cr | Average 11.38 LPA | Scholarships Available
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q2 |
71.85 |
Electrostatics |
Gauss's Law and Dielectrics |
|
Q6 |
66.47 |
Optics |
Reflection by Spherical Mirrors |
|
Q3 |
54.09 |
Rotational Motion |
Angular SHM of a Rigid Body |
|
Q8 |
48.78 |
Thermodynamics |
Second Law of Thermodynamics |
Question 2: Two co-axial conducting cylinders of same length $\ell$ with radii $\sqrt{2} R$ and $2 R$ are kept, as shown in Fig. 1. The charge on the inner cylinder is $Q$ and the outer cylinder is grounded. The annular region between the cylinders is filled with a material of dielectric constant $\kappa=5$. Consider an imaginary plane of the same length $\ell$ at a distance $R$ from the common axis of the cylinders. This plane is parallel to the axis of the cylinders. The cross-sectional view of this arrangement is shown in Fig. 2. Ignoring edge effects, the flux of the electric field through the plane is $\left(\epsilon_0\right.$ is the permittivity of free space):
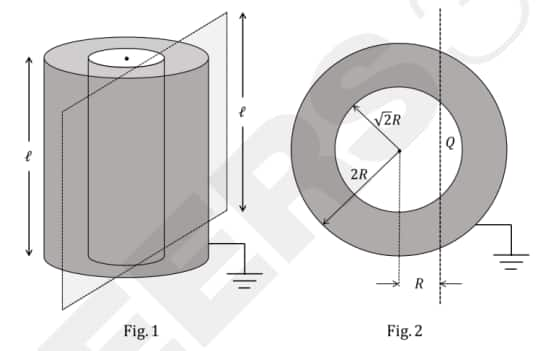
(1) $\frac{Q}{30 \epsilon_0}$
(2) $\frac{Q}{15 \epsilon_0}$
(3) $\frac{Q}{60 \epsilon_0}$
(4) $\frac{Q}{120 \epsilon_0}$
Solution:
Inner Radius: $r_{in} = \sqrt{2}R$
Outer Radius: $r_{out} = 2R$
Dielectric Constant: $\kappa = 5$
Plane Position: $x = R$ (vertical chord)
The flux passes through the plane only in the region where the electric field exists ($r_{in} \leq r \leq r_{out}$).
At inner boundary: $\cos \phi_1 = \frac{R}{\sqrt{2}R} = \frac{1}{\sqrt{2}} \implies \phi_1 = 45^\circ$
At outer boundary: $\cos \phi_2 = \frac{R}{2R} = \frac{1}{2} \implies \phi_2 = 60^\circ$
The angle subtended by the plane within the field region (on one side of the axis) is:
$\Delta \phi = 60^\circ - 45^\circ = 15^\circ$
Total angle for both sides: $30^\circ$
Total flux from the cylinder in the dielectric is $\Phi_{total} = \frac{Q}{\kappa \epsilon_0}$.
The plane captures a fraction of this flux based on its angle relative to the full $360^\circ$:
$\Phi_{plane} = \left( \frac{30^\circ}{360^\circ} \right) \frac{Q}{5\epsilon_0}$
$\Phi_{plane} = \frac{1}{12} \times \frac{Q}{5\epsilon_0} = \frac{Q}{60\epsilon_0}$
Hence, the correct answer is option (3)
Refer to the points given below to know why most students left this question unattempted:
1. In this question, students need to find the flux through a flat plane placed inside a cylindrical field. But students assume that they need to perform a gruelling double integral ($\int \vec{E} \cdot d\vec{A}$) over the rectangular plane.
2. Students sometimes forget to include $\kappa$ in the denominator of the flux formula, which leads to errors and miscalculations.
3. Calculation of specific angles ($45^\circ$ and $60^\circ$) using the radii $\sqrt{2}R$ and $2R$ becomes tedious and lengthy.
Refer to the JEE Advanced previous year question papers given below to know the expected toughest question of JEE Advanced Physics, overall difficulty level and the type of questions asked:
On Question asked by student community
Hi,
You can use the following link to predict your college options. https://engineering.careers360.com/jee-advanced-college-predictor
Hope it helps!
Hello,
With a CRL rank of 5,13,695 and SC rank of 38,157 , getting top NITs or popular branches may be difficult. However, you can still consider some GFTIs and lower-demand branches in newer NITs.
You may look at:
Assam University
Sant Longowal Institute of Engineering and Technology
Some newer
Hello,
With a JEE Advanced rank of around 27,300 , getting an IIT seat in the General category is quite difficult, as most IIT seats close at lower ranks. However, you may still have a chance in some newer IITs and less competitive branches, depending on your category and gender.
Hello,
Yes, with 62 marks in JEE Advanced 2026 and SC category , you do have a chance of getting an IIT seat.
The official qualifying cutoff for the SC category is 46 marks , and you've scored well above that, which means you should get a rank in the
IIT Jammu, IIT Tirupati, IIT Palakkad)
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Top Placements: 50 LPA in Google | 46.38 LPA in Amazon | 45 LPA in Adobe | 50 LPA in Microsoft | 44.14 in Amazon
40 LPA Highest Package | Up to 100% Scholarship worth 24 Crore via GUTS exam
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score
Integrated M.Tech admissions open @ VIT Bhopal University | Highest CTC 70 LPA | Application Closing Soon | Apply now