UPES B.Tech Admissions 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
JEE Advanced Chemistry 2025 Analysis: Do you want to know about the type of questions asked in the Chemistry section of the JEE Advanced 2025? Want to know whether the paper was difficult or doable for most aspirants? Understanding these aspects can help students preparing for the next attempt plan their strategy more effectively. The JEE Advanced Chemistry 2025 paper checks students conceptual clarity, analytical thinking, and application of fundamental principles from Physical, Organic, and Inorganic Chemistry. Through this JEE Advanced Chemistry 2025 Analysis, we will examine the difficulty level of the paper, the important topics covered, and the overall question trends.
This Story also Contains

Students preparing for JEE Advanced can benefit greatly from analysing previous exam papers. Identification of types of questions that are frequently skipped, wrongly attempted, or solved correctly gives a clear idea of how students performed and shows which parts of the paper were the toughest in JEE Advanced 2025 Chemistry.
Chemistry of JEE Advanced 2025 in both Paper 1 and Paper 2 is a perfect blend of simple and tricky questions. Given below a closer look at candidate wise performance on which types of questions were solved accurately, skipped frequently, or answered incorrectly from Chemistry.
|
Paper 1/2 |
Question (2025) |
Not Attempted |
% Not Attempted |
Full Marks |
% Full Marks |
Partial Marks |
% Partial Marks |
Wrong Response |
|
1 |
Q.1 |
69494 |
38.52 |
61228 |
33.94 |
49700 | ||
|
1 |
Q2 |
49702 |
27.55 |
85090 |
47.16 |
45630 | ||
|
1 |
Q3 |
42763 |
23.7 |
81216 |
45.01 |
56443 | ||
|
1 |
Q4 |
40324 |
22.35 |
54822 |
30.39 |
85276 | ||
|
1 |
Q5 |
26693 |
14.79 |
29921 |
16.58 |
26305 |
14.58 |
97503 |
|
1 |
Q6 |
60319 |
33.43 |
50456 |
27.97 |
30410 |
16.85 |
39237 |
|
1 |
Q7 |
59779 |
33.13 |
70100 |
38.85 |
0 |
0 |
50543 |
|
1 |
Q8 |
31189 |
17.29 |
46128 |
25.57 |
103105 | ||
|
1 |
Q9 |
39526 |
21.91 |
2295 |
1.27 |
138601 | ||
|
1 |
Q10 |
68500 |
37.97 |
4623 |
2.56 |
107299 | ||
|
1 |
Q11 |
39742 |
22.03 |
7372 |
4.09 |
133308 | ||
|
1 |
Q12 |
61356 |
34.01 |
21739 |
12.05 |
97327 | ||
|
1 |
Q13 |
73051 |
40.49 |
871 |
0.48 |
106500 | ||
|
1 |
Q14 |
64795 |
35.91 |
92876 |
51.48 |
22751 | ||
|
1 |
Q15 |
21895 |
12.14 |
99348 |
55.06 |
59179 | ||
|
1 |
Q16 |
63560 |
35.23 |
90312 |
50.06 |
26550 | ||
|
2 |
Q1 |
50927 |
28.23 |
54561 |
30.24 |
74934 | ||
|
2 |
Q2 |
58469 |
32.41 |
56427 |
31.28 |
65526 | ||
|
2 |
Q3 |
62405 |
34.59 |
35693 |
19.78 |
82324 | ||
|
2 |
Q4 |
42895 |
23.77 |
50535 |
28.01 |
86992 | ||
|
2 |
Q5 |
76736 |
42.53 |
5729 |
3.18 |
33946 |
18.81 |
64011 |
|
2 |
Q6 |
14785 |
8.19 |
103996 |
57.64 |
30163 |
16.72 |
31478 |
|
2 |
Q7 |
79874 |
44.27 |
28321 |
15.7 |
19615 |
10.87 |
52612 |
|
2 |
Q8 |
60685 |
33.64 |
23533 |
13.04 |
27866 |
15.44 |
68338 |
|
2 |
Q9 |
51822 |
28.72 |
18287 |
10.14 |
110313 | ||
|
2 |
Q10 |
53775 |
29.81 |
12980 |
7.19 |
113667 | ||
|
2 |
Q11 |
64244 |
35.61 |
5663 |
3.14 |
110515 | ||
|
2 |
Q12 |
51706 |
28.66 |
20645 |
11.44 |
108071 | ||
|
2 |
Q13 |
44997 |
24.94 |
14813 |
8.21 |
120612 | ||
|
2 |
Q14 |
36717 |
20.35 |
3915 |
2.17 |
139790 | ||
|
2 |
Q15 |
8324 |
4.61 |
42441 |
23.52 |
129657 | ||
|
2 |
Q16 |
55074 |
30.53 |
32631 |
18.09 |
92717 |
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Some JEE Advanced Chemistry questions were left unanswered. Analysing these non attempt questions helps identify the topics that are difficult to understand.
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q13 |
40.49 |
Aldehydes, Ketones, And Carboxylic Acid |
Williamson's ether synthesis |
|
Q10 |
37.97 |
States Of Matter |
Vander Waals equation |
|
Q14 |
35.91 |
P- Block Elements |
Salt analysis |
|
Q16 |
35.23 |
Amines |
Tests for functional groups |
|
Q12 |
34.01 |
Organic chemistry some basic principles and techniques |
Nitrogen estimation and carbylamine test |
|
Question Number |
% Not Attempted |
Chapter Name |
Concept Name |
|
Q7 |
44.27 |
Amines |
Gabriel phthalamide synthesis |
|
Q5 |
42.53 |
States Of Matter |
intermolecular forces |
|
Q11 |
35.61 |
Surface Chemistry |
Freundlich adsorption isotherm |
|
Q3 |
34.59 |
Aldehydes, Ketones And Carboxylic Acid |
Named reactions Aldol condensation, oxymercuration, ozonolysis |
|
Q8 |
33.64 |
Aldehydes, ketones and carboxylic acids |
Reduction, oxidation, dehydration reactions |
Question 7 of Chemistry JEE Advanced is one of the key problems that tested both conceptual clarity and application skills

Solution:
Oxidation
Tetralin on strong oxidation with acidic permanganate forms phthalic acid, which on heating with ammonia forms phthalimide.
$\text{Tetralin} \xrightarrow[ \Delta]{KMnO_4/H^+} C_6H_4(COOH)_2$
$C_6H_4(COOH)_2 + NH_3 \xrightarrow{\Delta} C_6H_4(CO)_2NH + 2H_2O$
Thus,
$X = \text{Phthalimide}$
Gabriel synthesis
Deprotonation and alkylation:
$C_6H_4(CO)_2NH + KOH \rightarrow C_6H_4(CO)_2N^-K^+ + H_2O$
$C_6H_4(CO)_2N^-K^+ + RBr \rightarrow C_6H_4(CO)_2NR$
$Y = \text{N-alkyl phthalimide}$
Hydrolysis
$C_6H_4(CO)_2NR + NaOH \rightarrow C_6H_4(COONa)_2 + RNH_2$
$Z = RNH_2$
(A) $X$ and $Y$ contain carbonyl groups → True
(B) Carbylamine reaction:
$
RNH_2 + CHCl_3 + 3KOH \rightarrow RNC + 3KCl + 3H_2O
$
Occurs only for primary amines, not $Y$ → False
(C) $Z = RNH_2$ reacts with Hinsberg reagent → True
(D) $Z$ is aromatic primary amine → False
$\boxed{A \text{ and } C}$
Hence, the correct answers are option 1,3.
1. First step to solve this question is Multi step Conversion where the reaction required linking oxidation → phthalimide formation → Gabriel synthesis → hydrolysis.
2. The key intermediate here is phthalimide, which is not a very common compound in everyday practice questions. Since many students had not memorized that naphthalene oxidation → phthalic acid → phthalimide, they struggled to identify “X” confidently.
3. In this question Option (B) tempted students with the carbylamine test, but the intermediate Y is not a primary amine. While option (D) suggested Z is an aromatic amine, but Gabriel synthesis doesn’t yield aryl amines.
4. This questions was placed among organic reaction sequence questions, which are usually time-consuming. Many aspirants skipped it to avoid losing time on an unfamiliar, lengthy pathway, preferring quicker physical or inorganic chemistry questions.
Some Chemistry JEE Advanced 2025 questions have a high percentage of wrong responses, which shows common misconceptions and calculation errors among candidates. Analysing these questions helps students understand the tricky concepts.
|
Question Number |
% Wrong Response |
Chapter Name |
Concept Name |
|
Q9 |
76.82 |
Equilibrium |
Weak acid dissociation |
|
Q11 |
73.89 |
Thermodynamics |
Expansion work of ideal gas |
|
Q10 |
59.47 |
States Of Matter |
Vander waals equation |
|
Q13 |
59.03 |
Aldehydes, Ketones And Carboxylic Acid |
Williamsons ether synthesis |
|
Q8 |
57.15 |
Electrochemistry |
Electrolysis and faradays law |
|
Question Number |
% Wrong Response |
Chapter Name |
Concept Name |
|
Q14 |
77.48 |
Electrochemistry |
Relation between gibbs free energy and cell potential |
|
Q15 |
71.86 |
Coordination Compounds |
Crystal field theory |
|
Q13 |
66.85 |
Solutions |
Osmotic pressure |
|
Q11 |
61.25 |
Surface Chemistry |
Freundlich adsorption isotherm |
|
Q12 |
59.9 |
Chemical Kinetics |
Pseudo-first-order reaction |
Question 14: An electrochemical cell is fueled by the combustion of butane at 1 bar and 298 K. Its cell potential is $\frac{X}{F} \times 10^3$ volts, where $F$ is the Faraday constant. The value of $X$ is $\_\_\_\_$.
Use : Standard Gibbs energies of formation at 298 K are :
$\begin{aligned}
& \Delta_f G_{\mathrm{CO}_2}^{\mathrm{o}}=-394 \mathrm{~kJ} \mathrm{~mol}^{-1} ; \\
& \Delta_f G_{\text {water }}^{\mathrm{o}}=-237 \mathrm{~kJ} \mathrm{~mol}^{-1} ; \Delta_f G_{\text {butane }}^{\mathrm{o}}=-18 \mathrm{~kJ} \mathrm{~mol}^{-1}
\end{aligned}$
Solution:
Given
1. Combustion reaction of butane
$C_4H_{10} + \frac{13}{2}O_2 \rightarrow 4CO_2 + 5H_2O$
2. Standard Gibbs free energy change
$\Delta G^\circ = \sum \Delta_f G^\circ(\text{products}) - \sum \Delta_f G^\circ(\text{reactants})$
$\Delta G^\circ = [4(-394) + 5(-237)] - [(-18)]$
$\Delta G^\circ = (-1576 - 1185) + 18$
$\Delta G^\circ = -2743 \ \text{kJ mol}^{-1}$
3. Number of electrons transferred
Carbon oxidation state change:
$-2.5 \rightarrow +4$
Increase per carbon:
$4 - (-2.5) = 6.5$
For 4 carbons:
$n = 4 \times 6.5 = 26$
4. Cell potential relation
$\Delta G^\circ = -nFE^\circ$
$E^\circ = \frac{2743 \times 10^3}{26F}$
$E^\circ = \frac{105.5}{F} \times 10^3$
$\boxed{X = 105.5}$
Hence, the correct answer is 105.5
1. Combination of Gibbs energy calculations with electrochemistry concepts is asked here. Finding and applying the link between $\Delta \mathrm{G}^0$ of reaction and cell potential can be difficult.
2. Students often do mistakes in Balancing of reactions or Electron Count
3. Errors may arise in the step where $\Delta \mathrm{G}^0$ for the overall reaction is calculated using standard Gibbs free energies of formation.
4. This question involves multiple calculation steps like reaction balancing → $\Delta \mathrm{G}^0$ → electrons transferred → unit conversions, under time constraints students tend to skip detailed re-checks. Any small slip along the way compounds to an incorrect final value.
Some JEE Advanced 2025 Chemistry questions were answered correctly by the majority of candidates, showing clear understanding and strong command over fundamental concepts.
|
Question Number |
% Full Marks |
Chapter Name |
Concept Name |
|
Q15 |
55.06 |
Amines |
Named reactions |
|
Q14 |
51.48 |
P- Block Elements |
Salt analysis |
|
Q16 |
50.06 |
Amines |
Tests for functional groups |
|
Q2 |
47.16 |
Coordination Compounds |
Spectrochemical series and electronic transition |
|
Q3 |
45.01 |
P- Block Elements |
Redox reactions of $\mathrm{KMnO}_4$ |
|
Question Number |
% Full Marks |
Chapter Name |
Concept Name |
|
Q6 |
57.64 |
P- Block Elements |
oxyacids of phosphorus |
|
Q2 |
31.28 |
P- Block Elements |
Hydrolysis of interhalogens |
|
Q1 |
30.24 |
Coordination compounds |
Sodium nitroprusside test |
|
Q4 |
28.01 |
Alcohols, Phenols And Ether |
oxidative clevage |
|
Q15 |
23.52 |
Coordination Compounds |
Crystal field theory |
Below is the total marks distribution in Chemistry JEE Advanced 2025 that shows how candidates performed overall, including those who qualified and those who were allotted seats.
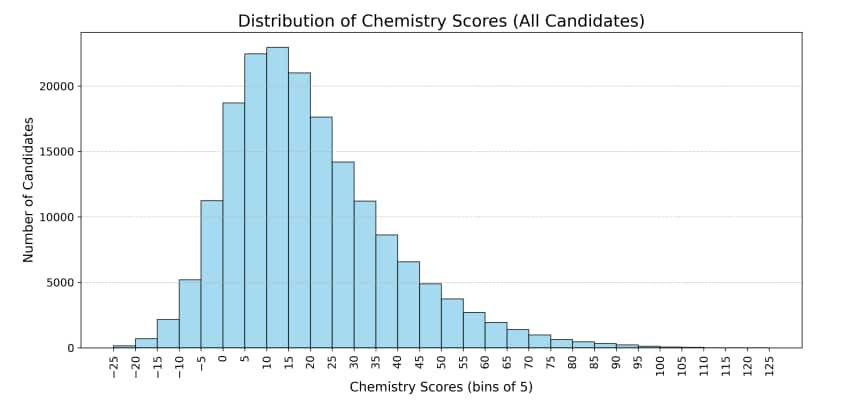
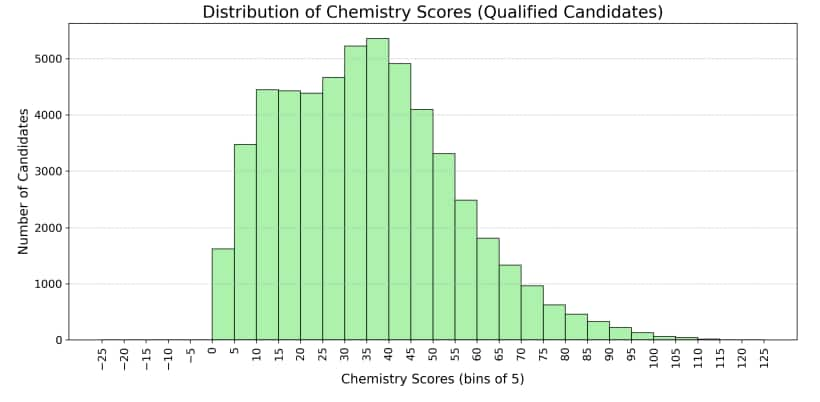
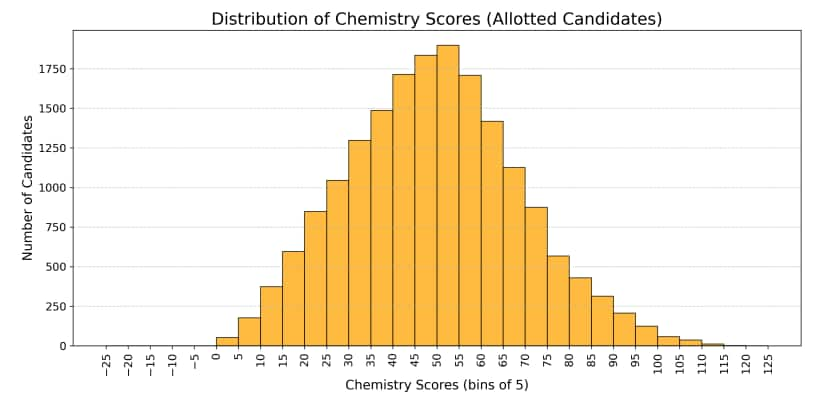
Some Chemistry questions in JEE Advanced 2025 were difficult. These questions had high wrong response or non attempt rates.
|
Q. No |
% Not Attempted |
% Full Marks |
% Wrong Response |
Chapter |
Concept |
|
Q13 |
40.49 |
0.48 |
59.03 |
Aldehydes, Ketones And Carboxylic Acid |
Williamsons ether synthesis |
|
Q11 |
22.03 |
4.09 |
73.89 |
Thermodynamics |
Expansion work of ideal gas |
|
Q10 |
37.97 |
2.56 |
59.47 |
States Of Matter |
Vander waals equation |
|
Q9 |
21.91 |
1.27 |
76.82 |
Equilibrium |
Weak acid dissociation |
The most difficult questions in JEE Advanced chemistry Paper 1 are Q13, Q11, Q10, Q9. Questions 9 and 11 have, on average, 75.35% wrong attempts and Question 13 is the least full-mark (0.48%) question.
|
Q. No |
% Not Attempted |
% Full Marks |
% Wrong Response |
Chapter |
Concept |
|
Q1 |
28.23 |
30.24 |
41.53 |
Coordination compounds |
Sodium nitroprusside test |
|
Q2 |
32.41 |
31.28 |
36.32 |
P- Block Elements |
Hydrolysis of interhalogens |
|
Q8 |
33.64 |
13.04 |
37.88 |
Aldehydes, ketones and carboxylic acids |
Reduction, oxidation, dehydration reactions |
|
Q4 |
23.77 |
57.64 |
48.22 |
Alcohols, Phenols And Ether |
oxidative cleavage |
Students preparing for the JEE Advanced often analyse the question trends of JEE Main, as both exams test conceptual understanding in Chemistry. This JEE Main 2026 and 2025 paper analysis helps students identify commonly asked topics, understand the difficulty level, and type of questions that frequently appear in exams.
Analysis of JEE Main 2026 Session 1
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Analysis of JEE Main 2025
Frequently Asked Questions (FAQs)
The top skipped questions often include those that are conceptually challenging or require intricate calculations. In 2025, many candidates tended to skip questions related to coordination chemistry and complex reaction mechanisms. Such questions demand not only understanding but also the ability to apply this knowledge under time constraints.
Yes, certain topics showed a higher incidence of wrongly attempted questions. Organic chemistry, particularly reaction mechanisms and stereochemistry, was a common hurdle for many students. The complexity of these concepts can lead to confusion and mistakes, especially if a student misapplies a rule or doesn't fully grasp the underlying principles.
Candidates performed particularly well on questions that involved stoichiometric calculations and basic principle-based questions. These topics are generally more straightforward and align closely with the foundational concepts taught in many chemistry courses. Questions requiring direct applications of laws, like the mole concept or basic thermodynamics, were often answered correctly, suggesting that students had a strong grasp of these fundamental ideas.
To improve performance, candidates should focus on enhancing their conceptual understanding through targeted study. Practicing past years’ papers and taking mock tests can help identify weak areas. Additionally, forming study groups or seeking help from mentors can clarify complex concepts. It’s also vital for students to time themselves during practice to simulate exam conditions.
Students can adopt several strategies to tackle complex topics. First, breaking down challenging concepts into smaller, manageable parts makes them easier to comprehend. Using visual aids, like charts and diagrams, can help in understanding intricate processes. Additionally, practicing with varied problems and scenarios reinforces knowledge.
On Question asked by student community
Hello,
With COMEDK rank 510 , JEE Main rank 40,989 , and JEE Advanced rank 28,900 , your best strategy is:
COMEDK first priority – You have a very good rank and can target top colleges like RVCE, BMSCE, and MSRIT.
JoSAA counselling – Fill NITs, IIITs, and GFTIs according
Hi,
You can use the following link to predict your college options. https://engineering.careers360.com/jee-advanced-college-predictor
Hope it helps!
Hello,
With a CRL rank of 5,13,695 and SC rank of 38,157 , getting top NITs or popular branches may be difficult. However, you can still consider some GFTIs and lower-demand branches in newer NITs.
You may look at:
Assam University
Sant Longowal Institute of Engineering and Technology
Some newer
Hello,
With a JEE Advanced rank of around 27,300 , getting an IIT seat in the General category is quite difficult, as most IIT seats close at lower ranks. However, you may still have a chance in some newer IITs and less competitive branches, depending on your category and gender.
Hello Dear Student,
Yes, having a JEE Advanced Preparatory Rank (e.g., 303) means you have a solid chance of securing a seat in an IIT Preparatory Course through JoSAA counseling.
You can check, find and access more information here:
https://engineering.careers360.com/articles/jee-advanced-rank-wise-colleges-list
Hope it helps!
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
40 LPA Highest Package | Up to 100% Scholarship worth 24 Crore via GUTS exam
3000+ Successful Placements | 100+ Leading Recruiters
Top Placements: 50 LPA in Google | 46.38 LPA in Amazon | 45 LPA in Adobe | 50 LPA in Microsoft | 44.14 in Amazon
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score