Amity University Noida-B.Tech Admissions 2026
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
JEE Main Chemistry sample paper with answer key - To prepare well for chemistry in the JEE Main 2026 exam, it is necessary first to understand what type of content to study, the exam format, and what kinds of questions will be asked. To assist you in your chemistry preparation, we have provided you with a sample paper for JEE Main 2026. This JEE Main 2026 Chemistry sample paper with answer key is designed based on the new NTA format, and will simulate the types of questions you may see in the test and help you assess how well you know the concepts and how quickly you can solve problems.
This Story also Contains

Along with the sample paper of JEE Main 2026, you will receive a complete answer key of chemistry, which includes detailed solutions for each question, enabling you to check your performance and understand how to answer each question correctly. In the next sections, you will find the link to the entire sample paper, and you can download the sample paper to practice. Now, let us get started with the JEE Main 2026 Chemistry sample paper with the answer key for april session.
Also Read: JEE Main 2026 April Attempt Strategy
The JEE Main 2026 Chemistry Sample Paper with Answer Key is created to help students practice exam-oriented questions and improve their understanding of both Physical, Organic, and Inorganic Chemistry. We are also giving the direct link so you can access, download, and solve the sample paper with ease.
For additional JEE Mains Chemistry Sample Papers with Solutions 2026, students can use the free Careers360 resource. It provides mock tests, sample papers, chapter-wise and subject-wise tests, plus JEE Main Free Study Material.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
1. Simulating Actual Examination Conditions
Simulating actual Examination Conditions by solving the JEE Main 2026 Chemistry sample paper with answer key using a timer similar to the one used in the actual JEE Main Examination helps to develop speed, accuracy, and examination temperament.
2. Identifying Your Weak Chapters
After attempting the Sample Papers, look at the topics that you performed poorly on, such as Chemical Bonding, Thermodynamics, Organic Reaction Mechanisms, etc., and revise those specific chapters before proceeding to the next set of Sample Papers.
3. Understanding Why Your Answers Were Correct or Incorrect
Go through the JEE Mains Chemistry Sample Paper with Solutions 2026 to fully understand the rationale behind each answer, whether it was correct or incorrect. Understanding why you answered questions incorrectly will help you avoid making the same mistakes again.
4. Creating Notes of Formulas & Reactions
While going through the JEE Main Chemistry sample paper with answer key 2026, make sure to go through the important formulas of chemistry, patterns in reactions, exceptions, or shortcut techniques in your notes.
5. Monitoring Your Improvement
While practising with the JEE Main Chemistry sample paper with answer key 2026, continue to take at least one or two Sample Papers each week and compare your scores to determine your continued improvement and develop self-confidence.
We have given some questions from the chemistry sample paper for reference.
Question 1: Identify the correct statement among the following:
Option 1: All naturally occurring amino acids except glycine contain one chiral centre. Option 2: All naturally occurring amino acids are optically active.
Option 3: Glutamic acid is the only amino acid that contains a –COOH group at the side chain.
Option 4: Amino acid cysteine easily undergoes dimerisation due to the presence of the free SH group.
Correct Answer: Amino acid, cysteine, easily undergoes dimerisation due to the presence of a free SH group.
Solution:
* Isoleucine has 2 chiral centres
* Glycine is optically inactive
* Aspartic acid also contains COOH groups at the side chain.
* Cysteine easily dimmerise due to the free SH group
Question 2: Consider the following molecules :
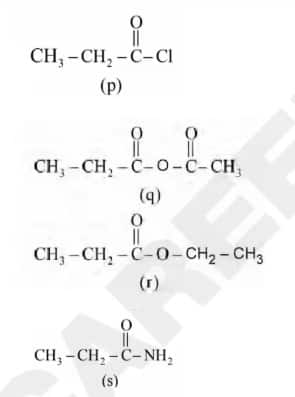
The correct order of the rate of hydrolysis is
Option 1: r >q>p>s
Option 2: q >p>r>s 41
Option 3: p >r>q>s
Option 4: p >q>r>s Correct
Answer: p >q>r>s
Solution:
Rate of hydrolysis ∝ Leaving group ability
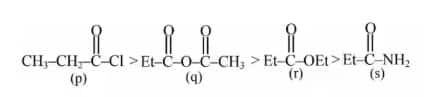
Hence, the correct answer is option (4).
Question 3: A solution is made by mixing one mole of volatile liquid A with 3 moles of volatile liquid B. The vapour pressure of pure A is 200 mm Hg and that of the solution is 500 mm Hg. The vapour pressure of pure B and the least volatile component of the solution, respectively, are :
Option 1: 1400 mm Hg, A
Option 2: 1400 mm Hg, B
Option 3: 600 mm Hg, B
Option 4: 600 mm Hg, A
Correct Answer: 600 mm Hg, A
Solution:
$P_S=P_A^{\circ} \cdot X_A+P_B^{\circ} \cdot X_B$
$500=200 \times \frac{1}{4}+P_B^{\circ} \cdot \frac{3}{4}$
$P_B^{\circ}=600 \text{ mmHg}$
$\text{As } P_A^{\circ} < P_B^{\circ} \Rightarrow \text{A is least volatile.}$
Hence, the correct answer is option (4).
Question 4: The property/properties that show irregularity in the first four elements of group-17 is/are :
(A) Covalent radius
(B) Electron affinity
(C) Ionic radius
(D) First ionisation energy
Choose the correct answer from the options given below:
Option 1: B and D only
Option 2: A and C only
Option 3: B only
Option 4: A, B, C and D
Correct Answer: B only
Solution:
The order of the first four elements of group 17 is as follows
F< Cl< Br< I (Covalent radius)
Cl > F>Br>I (Electron affinity)
$\mathrm{F}^{-}<\mathrm{Cl}^{-}<\mathrm{Br}^{-}<\mathrm{I}^{-}$
F >Cl>Br>I ( Ist ionization energy)
Electron affinity order is irregular.
Hence, the correct answer is option (3).
Frequently Asked Questions (FAQs)
Check accuracy, time spent per question, types of mistakes that can be conceptual or calculation, and topics where you repeatedly lose marks.
Yes, majorly the sample papers are made following the latest NTA exam pattern, which includes MCQs and numerical value-type questions.
On Question asked by student community
Hello,
With 93 percentile in JEE Main , 34,075 rank in TS EAPCET , and 92% in IPE , you have a decent profile for Category-B (Management Quota) admission at MGIT.
Getting CSE through regular counselling is difficult with a 34k rank, but through the B Category quota , you
Hello,
Yes, with 62 marks in JEE Advanced 2026 and SC category , you do have a chance of getting an IIT seat.
The official qualifying cutoff for the SC category is 46 marks , and you've scored well above that, which means you should get a rank in the
IIT Jammu, IIT Tirupati, IIT Palakkad)
Hello,
If 8185 is your SC category rank , getting a seat at IIT Dharwad may be difficult, especially in the more popular branches. However, it's still worth participating in JoSAA counselling because cutoffs can vary from year to year, and sometimes seats are available in later rounds.
If you're
Hello,
If you get a seat through the IIT preparatory course , I would generally suggest taking it rather than dropping a year.
A drop year always comes with uncertainty. Even if you prepare well, there's no guarantee that your rank will improve enough next year. On the other hand,
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Top Placements: 50 LPA in Google | 46.38 LPA in Amazon | 45 LPA in Adobe | 50 LPA in Microsoft | 44.14 in Amazon
40 LPA Highest Package | Up to 100% Scholarship worth 24 Crore via GUTS exam
B.Tech Admissions 2026 Open | Get Career Support, Mock interviews, Soft skill training
Affiliated with Visvesvaraya Technological University (VTU) | Merit-based admission process