Amity University Noida-B.Tech Admissions 2026
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
JEE Main Chemistry Formulas 2027 - The Joint Entrance Exam is one of the most competitive engineering entrance exams. It is divided into three subsections (Physics, Chemistry, and Maths). Chemistry is an important subject for JEE Main preparation. Hence, having a list of JEE Main Chemistry Formulas 2027 is necessary. Every aspirant should have the short notes for JEE Main 2027 that comprise the important formulas. These important JEE formulas for Chemistry help in solving the typical problems easily.
This Story also Contains

There will be a total of 25 questions from chemistry in JEE Main 2027, all of which have to be attempted. It is further segregated into Physical, Organic, and Inorganic sections. The maximum number of topics has been reduced from this section (chemistry) only. This makes Chemistry easier than before. However, the high-weightage topics for JEE Main remain the same.
Below are important formulas for JEE Main 2027 Chemistry that are helpful for both sessions. Candidates should solve as many chemistry questions as they can and implement these formulas to remember them quickly. To crack the JEE Main exam with good marks, paste these formulas near the study table and memorise them.
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Candidates, while studying chemistry, need to revise and practice the chemical equations and symbols. To some, chemistry is a tough subject, but when candidates practice chemical equations and revise the properties, formulas and symbols, they will have command over the subject. Candidates can check the JEE Main Chemistry formulas below
Some Basic Concepts in chemistry
1. Boyle's Law: $P_1 V_1=P_2 V_2$ (at constant T and n)
2. Charles's Law: $\frac{V_1}{T_1}=\frac{V_2}{T_2}$ (at constant P and n)
3. Avogadro's Law: $\frac{v}{n}=$ constant
4. Average Atomic Mass
$$
=\frac{\Sigma(\text { Mass of Isotopes })_i \times(\% \text { abundance })_i}{100}
$$
5. Mole $=\frac{\mathrm{W}}{\mathrm{M}}=\frac{\text { (Wt. of substance in gm.) }{ }^{100}}{\text { (Molar mass of substance(G.m.m)) }}$
6. Mass $1 \%$ of an element
$$
=\frac{\text { Mass of that element in one mole of the compound }}{\text { Molar mass of the compound }} \times 100
$$
7. Equivalent Weight $=\frac{\text { Molecular weight }}{n-\text { factor }(x)}$
Atomic Structure
1. Frequency $v=\frac{1}{T}$
2. Wave number $(\bar{v}) \bar{v}=\frac{1}{\lambda}$
3. $E=h v=\frac{h c}{\lambda}$
4. Line Spectrum of Hydrogen-like atoms
$$
\frac{1}{\lambda}=R Z^2\left(\frac{1}{n_1^2}-\frac{1}{n_2^2}\right)
$$
5. Bohr radius of nth orbit:
$$
\mathrm{r}_{\mathrm{n}}=0.529 \frac{\mathrm{n}^2}{\mathrm{z}} \mathrm{~A}^0
$$
6. Velocity of electron in nth orbit:
$$
V_n=\left(2.18 \times 10^6\right) \frac{\mathrm{Z}}{\mathrm{n}} \mathrm{~m} / \mathrm{s}
$$
where Z is atomic number
7. Total energy of electron in nth orbit:
$$
E_n=-13.6 \frac{Z^2}{n^2} \mathrm{eV}=-2.18 \times 10^{-18} \frac{Z^2}{n^2} \mathrm{~J}
$$
where Z is atomic number
8. Hisenberg Uncertainity Principle: $\Delta x \cdot \Delta P \geq \frac{h}{4 \pi}$
9. $\mathrm{E}_{\mathrm{n}}=-\frac{1312 \times \mathrm{Z}^2}{\mathrm{n}^2} \mathrm{~kJ} / \mathrm{mol}$
Chemical Thermodynamics
1. Expansion Work $=\mathrm{P} \times \Delta \mathrm{V}=-\mathrm{P}_{\text {ext. }}\left[\mathrm{V}_2-\mathrm{V}_1\right] \mathrm{P}=$ external pressure And $\Delta \mathrm{V}=$ increase or decrease in volume.
2. Work done in a reversible isothermal process
$$
\begin{aligned}
\mathrm{W} & =-2.303 \mathrm{nRT} \log _{10} \frac{\mathrm{~V}_2}{\mathrm{~V}_1} \\
\mathrm{~W} & =-2.303 \mathrm{nRTlog} \frac{\mathrm{P}_1}{\mathrm{P}_2}
\end{aligned}
$$
3. Work done in an irreversible isothermal process
$$
\text { Work }=-P_{\text {ext. }}\left(V_2-V_1\right)
$$
That is, Work $=-\mathrm{P} \times \Delta \mathrm{V}$
4. $W=\Delta E=n C_V \Delta T$
5. Enthalpy: $H=U+p V$
6. First Law of Thermodynamics: $\Delta U=q+W$
7. $\Delta \mathrm{G}=\Delta \mathrm{H}-\mathrm{T} \Delta(\mathrm{S})$
8. $\Delta G=-n F E$
Equilibrium
1. For a reaction:
$$
\mathrm{mA}+\mathrm{nB} \rightleftharpoons \mathrm{pC}+\mathrm{qD} \frac{\mathrm{~K}_{\mathrm{f}}}{\mathrm{~K}_{\mathrm{b}}}=\frac{[\mathrm{C}]^{\mathrm{p}}[\mathrm{D}]^{\mathrm{q}}}{[\mathrm{~A}]^{\mathrm{m}}[\mathrm{~B}]^{\mathrm{n}}}=\mathrm{K}_{\mathrm{c}}
$$
2. $\mathrm{pH}=-\log _{10}\left[\mathrm{H}^{+}\right]$
3. $\mathrm{k}_{\mathrm{W}}=\left[\mathrm{H}^{+}\right]\left[\mathrm{OH}^{-}\right]=10^{-14}$
4. $\mathrm{pH}=\mathrm{pK}_{\mathrm{a}}+\log _{10} \frac{\text { [Salt] }}{\text { Acid }}$
5. $\mathrm{pOH}=\mathrm{pK}_{\mathrm{b}}+\log _{10} \frac{[\text { Salt] }}{[\text { Base }]}$
ELECTROCHEMISTRY
1. $W=\frac{\text { Eit }}{96500}$
$$
\frac{E_1}{E_2}=\frac{M_1}{M_2} \text { or } \frac{W_1}{W_2}=\frac{Z_1}{Z_2}
$$
2.
$E_1=$ equivalent weight
$E_2=$ equivalent weight
W or M = mass deposited
3.
$$
\begin{gathered}
E_{\text {cell }} \text { or } E M F=\left[E_{\text {red }}(\text { cathode })-E_{\text {red }}(\text { anode })\right. \\
E_{\text {eell }}^{\circ} \text { or } E M F^{\circ} \\
=\left[E_{\text {red }}^{\circ}(\text { cathode })-E_{\text {red }}^{\circ}(\text { anode })\right]
\end{gathered}
$$
$$
\begin{aligned}
& \text { 4. } \mathrm{E}=\mathrm{E}^{\circ}-\frac{\mathrm{RT}}{\mathrm{nF}} \ln Q \\
& \text { 5. } \mathrm{xA}+\mathrm{yB} \rightarrow \mathrm{mC}+\mathrm{nD}
\end{aligned}
$$
The emf can be calculated as
$$
\text { Ecell }=E^{\circ} \text { cell }-\frac{0.059}{n} \log \frac{[\mathrm{C}]^{\mathrm{m}}[\mathrm{D}]^{\mathrm{n}}}{[\mathrm{~A}]^{\mathrm{x}}[\mathrm{~B}]^{\mathrm{y}}}
$$
6.
$$
\Lambda_{\mathrm{m}}=\kappa \times \frac{1000}{\mathrm{c}}
$$
7. $\Lambda_{\mathrm{eq}}=\frac{1000 \times \kappa}{\mathrm{N}}$
Solutions
1.
Mass \% of a component $=\frac{\text { Mass of the component in the solution }}{\text { Total mass of the solution }} \times 100$
2. Volume $\%$ of a component $=\frac{\text { Volume of the component }}{\text { Total volume of solution }} \times 100$
3. Mass by Volume $\%$ of a component $=\frac{\text { Total valume of solution }}{\text { Total volume of solution }} \times 100$
4. Parts per million
$$
=\frac{\text { Number of parts of the component }}{\text { Total number of parts of all components of the solution }} \times 10^6
$$
5. Mole fraction of a component
$$
=\frac{\text { Number of moles of the component }}{\text { Total number of moles of all the components }}
$$
6. Molarity: $(M)=\frac{\text { No. of Moles of Solutes }}{\text { Volume of Solution in Liters }}$
7. Molality: $(m)=\frac{\text { No. of Moles of Solutes }}{\text { Mass of solvent inkg }}$
$$
P_A=P_A^o X_A
$$
8. $\left(P_T\right)=P_A^o X_A+P_B^o X_B\left(P_B=P_B^o X_B\right)$
9. $\Delta \mathrm{T}_{\mathrm{b}}=\mathrm{K}_{\mathrm{b}} \times \frac{\mathrm{W}}{\mathrm{M}} \times \frac{1000}{\mathrm{~W}}$
10. $\Delta T_f=K_f \times \frac{w}{M} \times \frac{1000}{W}$
11. $\Pi=C R T$
$$
\mathrm{i}=\frac{\text { Observed number of solute particles }}{\text { Number of particles initially taken }}
$$
12.
$$
\mathrm{i}=\frac{\text { Observed value of colligative property }}{\text { Theoretical value of colligative property }}
$$
Chemical kinetics
1. Unit of average velocity $=\frac{\text { Unit of concentration }}{\text { Unit of time }}=\frac{\text { mole }}{\text { litre second }}=$ mole litre -1 second -1
2. $a A+b B \rightarrow c C+d D$
Rate w.r.t. $[\mathrm{A}]=-\frac{\mathrm{d}[\mathrm{A}]}{\mathrm{dt}} \times \frac{1}{\mathrm{a}}$
Rate w.r.t. $[B]=-\frac{d[B]}{d t} \times \frac{1}{b}$
Rate w.r.t. $[\mathrm{C}]=-\frac{\mathrm{d}[\mathrm{C}]}{\mathrm{dt}} \times \frac{1}{\mathrm{c}}$
Rate w.r.t. $[\mathrm{D}]=-\frac{\mathrm{d}[\mathrm{D}]}{\mathrm{dt}} \times \frac{\mathrm{c}}{\mathrm{d}}$
3. $\mathrm{R} \propto[\mathrm{A}]^{\mathrm{p}}[\mathrm{B}]^{\mathrm{q}}$
4. Unit of Rate Constant-
The differential rate expression forn th order reaction is as follows:
$$
\begin{gathered}
-\frac{d x}{d t}=k(a-x)^n \\
\text { ork }=\frac{d x}{(a-x)^n d t}=\frac{(\text { concentration })}{(\text { concentration })^n \text { time }}=(\text { conc. })^{1-n} \text { time }^{-1}
\end{gathered}
$$
5. For the first-order reaction,
$$
\begin{aligned}
& k=\frac{2.303}{t} \log \frac{[\mathrm{R}]_0}{[\mathrm{R}]} \\
& \text { 6. } t_1 / 2=\frac{0.693}{k}
\end{aligned}
$$
7. For any general nth order reaction it is evident that,
$$
\mathrm{t}_{\frac{1}{2}} \propto[\mathrm{~A}]_0^{1-\mathrm{n}}
$$
It is to be noted that the above formula is applicable for any general nth-order reaction except $\mathrm{n}=1$.
8. Arrhenius Equation: $\mathrm{k}=\mathrm{Ae}^{-\mathrm{Ea} / \mathrm{RT}}$
9. $\log \frac{\mathrm{K}_2}{\mathrm{~K}_1}=\frac{\mathrm{Ea}}{2.303 \mathrm{R}}\left[\frac{1}{\mathrm{~T}_1}-\frac{1}{\mathrm{~T}_2}\right]$
Coordination Compounds
1. $$
\mathrm{EAN}=Z-O+2 L
$$
Where:
$Z=$ Atomic number of the central metal atom/ion
0= Oxidation state of the metal atom/ion
$\mathrm{L}=$ Number of ligands (or donor atoms) × number of electrons donated per ligand
2.
Crystal Field Stabilization Energy (CFSE):
Tetrahedral:
$$
\text { CFSE }=(-0.6 x+0.4 y) \Delta_t
$$
where $x=t_2 g$ electrons, $y=$ e g electrons
d- & f-Block Elements
1. Magnetic Moment:
$$
\mu=\sqrt{n(n+2)} \mathrm{BM}
$$
Chemical Bonding and Molecular Structure
1. Formal Charge:
$$
\text { F.C. }=V-N-\frac{B}{2}
$$
( $\mathrm{V}=$ valence electrons, $\mathrm{N}=$ non-bonding, $\mathrm{B}=$ bonding electrons)
2. Bond Order (Molecular Orbital Theory):
$$
\text { Bond Order }=\frac{\left(N_b-N_a\right)}{2}
$$
3. Dipole Moment:
$$
\begin{gathered}
\mu=q \times d \\
(q=\text { charge }, d=\text { distance between charges })
\end{gathered}
$$
Some Basic Principles of Organic Chemistry
1. Degree of Unsaturation (DU or IHD):
$$
\mathrm{DU}=\frac{2 C+2-H+N-X}{2}
$$
( $\mathrm{C}=$ carbon, $\mathrm{H}=$ hydrogen, $\mathrm{N}=$ nitrogen, $\mathrm{X}=$ halogen )
Hydrocarbons
1. Alkanes: $\mathrm{C}_n \mathrm{H}_{2 n+2}$
2. Alkenes: $C_n H_{2 n}$
3. Alkynes: $\mathrm{C}_n \mathrm{H}_{2 n-2}$
Carboxylic Acids and Derivatives
Method of Preparation of Carboxylic Acid
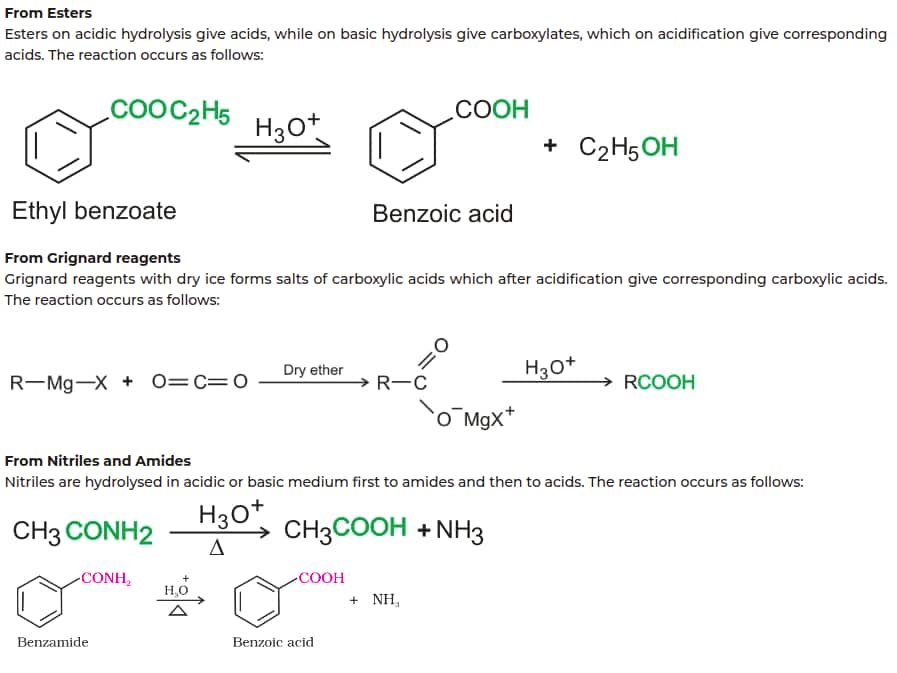
Not all Chemistry chapters are formula-heavy. Focus your JEE Main Chemistry Revision Notes on the chapters below—these have the most formulas to memorise and appear regularly in the exam pattern.
| Chapter | Section | Key Formulas |
| Thermodynamics | Physical | Hess's Law, Kirchhoff's equation |
| Chemical Kinetics | Physical | Rate law, Arrhenius equation, half-life formulas |
| Electrochemistry | Physical | Nernst equation, Faraday's laws, Kohlrausch's law |
| Chemical Equilibrium | Physical | Kc, Kp, relation between Kc & Kp, Henderson equation |
| Mole Concept & Stoichiometry | Physical | Molarity, molality, normality, equivalent concept |
| Gaseous State | Physical | Ideal gas law, Van der Waals equation, Graham's law |
| Atomic Structure | Physical | Bohr model, de Broglie, Rydberg formula |
| Solutions & Colligative Properties | Physical | Raoult's law, osmotic pressure, and elevation of boiling point |
| Coordination Compounds | Inorganic | EAN rule, crystal field splitting, and isomerism rules |
| Organic Name Reactions | Organic | Aldol, Cannizzaro, Sandmeyer, Reimer-Tiemann |
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score
Given below are some tips to help you prepare for JEE Main and score good marks in the exam:
1. First, students need to understand the JEE Main syllabus and Exam Pattern so that they get familiar with the syllabus and the exam pattern.
2. Try to find out the important and high-scoring topics of JEE Main Chemistry and prepare accordingly.
3. Make a good JEE Main study plan as per your preparation level. Divide your preparation in monthly, weekly and daily targets and spend more time on tough subjects or topics.
4. Students need to focus on the clarity of concepts; they need to know the logic behind the derivation of each formula.
5. Try to solve questions on a regular basis. Solve JEE Main previous year question papers and attempt mock tests and sample papers regularly.
Students find it difficult to learn formulas for JEE Main, but with the right approach, they can remember them. Given below are some points to remember:
1. Students must try to understand why a formula works and how chemical reactions occur, and their mechanism.
2. Then break down formulas into chapters or topics.
3. To learn these formulas easily, try to make a formula notebook.
4. Sometimes students must try to make mnemonics and short tricks, as it helps in quick revision.
5. Try to solve as many questions and revise
6. Try to use diagrams and flowcharts.
Small formula errors in Chemistry can cost easy marks in JEE Main 2027. Before your next mock test, make sure you are not making these mistakes with your JEE Main Chemistry Revision Notes.
Mixing sections—Keep Physical, Organic, and Inorganic Chemistry formulas in different sections of your Chemistry Formula Sheet PDF. Mixing them during the revision creates confusion in the exam.
Memorising without units—Formulas like the Nernst equation and Arrhenius equation have specific units. Always note them alongside the formula.
Ignoring NCERT derivations—Many Important Chemistry Formulas for JEE Main come directly from NCERT. If you only memorise the final expression, a slightly twisted question will trip you up.
Revising all chapters equally—The exam pattern shows some chapters appear far more than others. Revise based on weightage, not habit.
Frequently Asked Questions (FAQs)
Chemistry has three subparts (Physical, inorganic and organic). Physical chemistry has many numericals based on different formulas.
Yes, Class 12 syllabus carries more than 50% of weightage, so it is important for JEE Main Exam.
JEE Main is an exam conducted for those aspirants who want to take admission in NIITs, IITs and other engineering colleges, and it is also an eligibility test for JEE advance.
Physical Chemistry has the most formulas. Topics like Electrochemistry, Solutions, Chemical Kinetics, Thermodynamics, and Atomic Structure are heavily formula-based.
Not much. Organic Chemistry is mostly reaction and mechanism-based. The key formula to know is the Degree of Unsaturation and the general formulas for Alkanes, Alkenes, and Alkynes
Electrochemistry, Chemical Kinetics, Solutions, Thermodynamics, and Atomic Structure. These topics together make up a large portion of the Physical Chemistry questions in JEE Main and have many formulas.
For Inorganic Chemistry, yes. For Physical Chemistry, NCERT covers the basics, but practicing numerical problems is equally important.
Make a chapter-wise formula sheet and revise it daily. For Physical Chemistry, solve at least 2 to 3 numerical problems per formula. For Organic and Inorganic, focus more on reactions and NCERT revision.
On Question asked by student community
Hello Dear Student,
Yes, with an SC category rank of 31,370, you have a very strong chance of getting admission to IIITDM Kancheepuram. While highly demanding branches like Computer Science and Engineering may close earlier (around 3,400 - 92,000 depending on the round), your rank is highly competitive for other
Hello Dear Student,
With a JEE Main CRL of 1,22,306 and an OBC rank of 40,694, securing a seat in top-tier NITs or IIITs for core branches will be highly challenging. However, using your Gujarat Home State Quota, your best options include GFTIs, newer IIITs, state-funded universities, and reputable private
Hello,
With 982/1000 in IPE (98.2%) and 89 percentile in JEE Main , you have a good chance of getting a seat at SASTRA University , especially through the admission process that considers both Class 12 marks and JEE Main scores. Your excellent board marks will work strongly in your
Hello,
With a CRL rank of 1.57 lakh , OBC rank of 54,000 , and Haryana domicile , getting CSE at YMCA Faridabad is unlikely based on previous cutoff trends.
However, you may still have a chance in:
Electronics & Computer Engineering (depending on cutoff movement)
Hello Dear Student,
With an OBC-NCL category rank of 25,995 in JEE Main , your chances at the most sought-after branches in top IIITs are limited, but you still have several realistic options through JoSAA and especially CSAB Special Rounds .
You may have chances in:
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Top Placements: 50 LPA in Google | 46.38 LPA in Amazon | 45 LPA in Adobe | 50 LPA in Microsoft | 44.14 in Amazon
40 LPA Highest Package | Up to 100% Scholarship worth 24 Crore via GUTS exam
100+ Recruiters | 100% Placement Assistance | Scholarship Available for Meritorious Students
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score