Amity University Noida-B.Tech Admissions 2026
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
JEE Main 2026 Question Paper: Careers360 have uploaded all the JEE Main 2026 question papers. Candidates can now download all the JEE Main question paper for both the sessions and shifts. Using the JEE Mains 2026 question paper can help you understand the difficulty of the JEE Mains 2026 exam and can help you prepare for the JEE Main April session as well. The JEE Main April session is scheduled from 2 to 9 April, and all the question paper of the January session is updated in the article below. If you practice these January session question papers, they will significantly boost your preparation for the April session, and you'll get a clear idea of the paper pattern and level of difficulty.
This Story also Contains

The JEE Main 2026 question paper is an important resource for students who want to understand the exam pattern, difficulty level, and the types of questions asked in the examination. Analysing the question paper helps aspirants identify frequently asked topics and evaluate their overall performance after the exam. In this article, we provide the JEE Main 2026 question paper PDF, along with details about the exam pattern, subject-wise questions, and key highlights of the paper. Reviewing the JEE Main 2026 question paper can help students preparing for upcoming sessions improve their preparation strategy and focus on the most important areas of the syllabus.
JEE Mains 2026 question paper has been given below for Jan 21, Jan 22, Jan 23, Jan 24 and Jan 28, both shifts. You will be able to find questions with the answer Key below:
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
You can download the Shift wise question paper from the given table
The JEE Mains 2026 Jan 28 Shift 2 was concept-driven but scoring, with Physics being formula-based, Chemistry theory-focused (more inorganic), and Maths lengthy yet manageable. You can find detailed information in the JEE Main 2026 January 28 Shift 2 Question Paper PDF. You can find subject-wise detailed analysis given below:
| Subjects | Major Topics Asked | Difficulty Level |
|---|---|---|
| Physics | Modern Physics, Ray Optics, Formula-based numerical questions | Easy |
| Chemistry | Chemical Kinetics, Ionic Equilibrium (Buffer), Coordination Compounds, Amines, Inorganic-focused theory | Moderate |
| Mathematics | Vector & 3D Geometry, Integral Calculus, Differential Equations, Probability, Conic Sections, Trigonometry, Sequences & Series, Statistics | Moderate (Lengthy) |
| Overall | Statement-based questions across subjects; Maths lengthy; Physics formula-driven | Easy |
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score
JEE Mains 2026 Jan 28 Shift 2 Maths Questions
Q.1 If $\sum_{r=1}^{25} \frac{r}{r^4+r^2+1}=\frac{p}{q}$, where $p$ and $q$ are coprime positive integers, then $p+q$ is equal to:
1. 841
2. 976
3. 984
4. 890
Q.2 $\frac{6}{3^{26}}+\frac{10}{3^{25}}+\frac{10.2}{3^{24}}+\frac{10.2^2}{3^{23}}+\ldots+\frac{10.2^{24}}{3}$ is equal to
1. $2^{26}$
2. $2^{25}$
3. $3^{26}$
4. $3^{25}$
JEE Mains 2026 Jan 28 Shift 2 Physics Questions
Question 1:
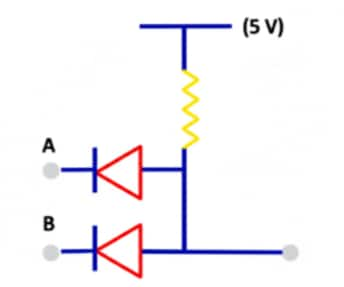
For the circuit given below, identify the logic gate.
1 AND
2 OR
3 NAND
4 NOR
JEE Mains 2026 Jan 28 Shift 2 Chemistry Questions
Question 1: The plot of $\log _{10} \mathrm{~K}$ vs $\frac{1}{T}$ gives a straight line. The intercept and slope, respectively, are
1) $\mathrm{c}=\log \mathrm{A}, \mathrm{m}=-\frac{E_a}{2.303 R}$
2) $\mathrm{c}=-\frac{E_a}{2.303 R^{\prime}}, \mathrm{m}=\log \mathrm{A}$
3) $\mathrm{c}=-\log \mathrm{A}, \mathrm{m}=-\frac{E_a}{2.303 R}$
4) $ \mathrm{c}=\log \mathrm{A}, \mathrm{m}=\frac{E_a}{2.303 R}$
The JEE Main 2026 Jan 28 morning shift (Shift 1) was moderate to tough. You can find detailed information in the JEE Main 2026 January 28 shift 1 Question Paper. For more details, you can check the table below:
| Subject / Overall | Difficulty | Length | Time | Question Type | Key Point |
|---|---|---|---|---|---|
| Physics | Easy–Moderate | Short–Moderate | ~60 min | Formula + Concept | Scoring and balanced |
| Mathematics | Moderate–Difficult | Lengthy | ~70–80 min | Calculation + Concept | Toughest and most time-consuming |
| Chemistry | Moderate | Moderate–Long | ~55–60 min | Statement-based + Named reactions | Organic-heavy but manageable |
| Overall Paper | Moderate–Hard | Lengthy | Time pressure | Statement-based trend | Maths toughest; Physics easiest |
Memory- Based Questions JEE Main 28 Shift 1
1. If $\alpha, \beta$ are roots of quadratic equation $\lambda x^2-(\lambda+3) x+3=0$ and $\alpha<\beta$ such that $\frac{1}{\alpha}-\frac{1}{\beta}=\frac{1}{3}$, then find sum of all possible values of $\lambda$.
1) 3
2) 2
3) 4
4) 6
2. Equation of an EMW in a medium is given by $E=2 \sin \left(2 \times 10^{15} t-10^7 x\right)$. Find the refractive index of the medium.
1) $\frac{3}{2}$
2) 2
3) $\frac{5}{3}$
4) $\frac{4}{3}$
The JEE Mains 2026 Jan 24 Shift 1 (morning shift). The overall difficulty level of the exam was Moderate. Few Memory-Based Questions
Question 1: An electron makes a transition from a higher energy orbit ( $\mathrm{n}_2$ ) to a lower energy orbit ( $\mathrm{n}_1$ ) in $\mathrm{Li}^2 {{+}}$ ion such that $\mathrm{n}_1+\mathrm{n}_2=4 \& \mathrm{n}_2+\mathrm{n}_1=2$. Determine the wavelength emitted in the dimension (in nm )
1) 12.9 nm
2) 11.4 nm
3) 16.7 nm
4) 9.2 nm
Question 2: Which of the following has the lowest bond energy?
$O_2^{+}, N_2^{+}, N_2^{-}$
Question 3: Choose the correct order of second IE of O, C, N and F
1) C $<$ N $<$ F $<$ O
2) $\mathrm{C}<\mathrm{F}<\mathrm{O}<\mathrm{N}$
3) $\mathrm{C}<\mathrm{N}<\mathrm{O}<\mathrm{F}$
4) C $<$ O $<$ F $<$ N
Question 4: Which of the following molecules is a secondary alcohol?
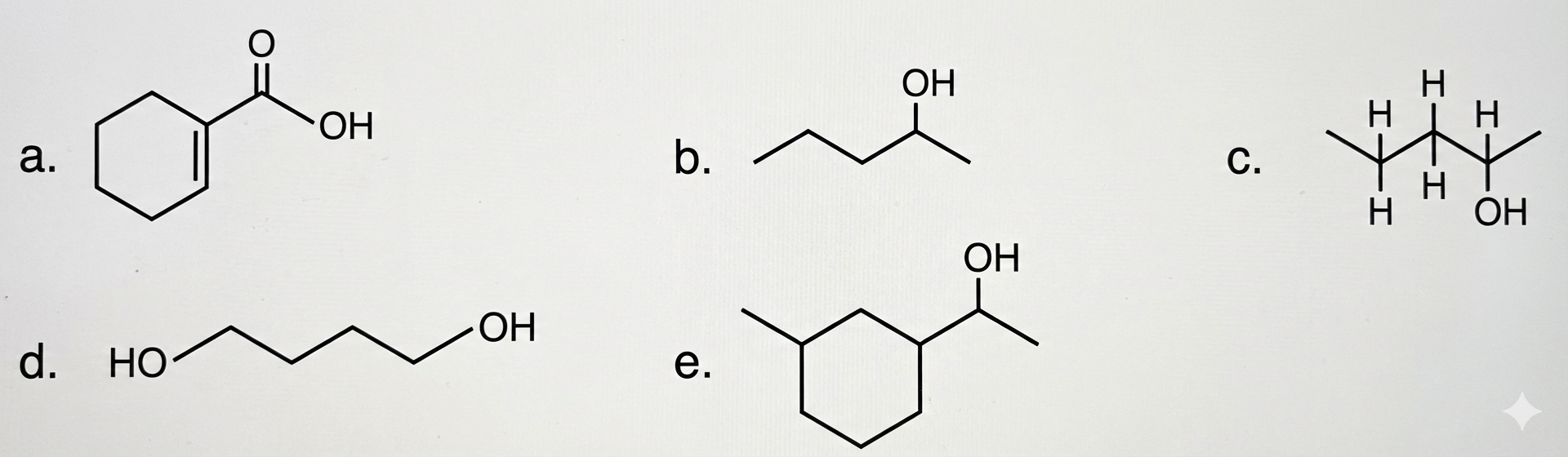
1 b, c, e only
2 b, c, d, e only
3 a, c, d, e only
4 a, b, d only
Compared to the 24th January JEE Main 2026 morning shift, this evening shift was almost similar. In terms of difficulty, the overall order was Chemistry > Mathematics> Physics. Few Memory-Based Questions
Question 1: statement-1: Two different aldehydes in cross-aldol condensation always give
four product :
statement-II: Among benzaldehyde and acetophenone, only acetophenone reacts with semicarbazine.
(1)Statement II and Statement II are both correct
(2) Statement I is incorrect. Statement II is correct
(3)Statement II is correct. Statement II is incorrect
(4) Statement-1 and Statement-II are both incorrect
Question 2: How many linear tripeptides are possible with valine (Val), Glycine (Gly) and Alanine (Ala)? No amino acid should be repeated?
1) 8
2) 5
3) 6
4) 4
Question 3: Which of the following have the same bond order and are paramagnetic?
1) $\mathrm{O}_2^{+}, \mathrm{N}_2^{-}$
2) $\mathrm{O}_2^{+}, \mathrm{O}_2^{-}$
3) $\mathrm{O}_2^{-}, \mathrm{N}_2^{-}$
4) $\mathrm{O}_2^{-}, \mathrm{N}_2^{+}$
The JEE Mains 2026 Jan 23 Shift 1 (morning shift) was easy to moderate. You can also check the JEE Main 2026 January 23 Shift 1 Question Paper.
| Subject | Difficulty Level | Key Observation |
|---|---|---|
| Physics | Easy | Formula-based, scoring |
| Chemistry | Moderate | Conceptual and statement-based |
| Mathematics | Moderate–Difficult | Lengthy, time-consuming calculations |
| Overall | Easy–Moderate | Comparable to JEE Main 2025 shifts |
Find some memory-based questions below:
Question 1: The correct order of ionisation energy of Cl, S, P, Al, Si is
1) Cl>P>S>Si>Al
2) P>Cl>S>Al>Si
3) Cl>S>P>Si>Al
4) Cl>Al>Si>P>S
Question 2: Given below are two statements
Statement-I : [CoBr4]2− absorbs lesser energy than [CoCl4]2−
Statement-II : [CoCl4]2− a has a higher crystal field splitting energy than [CoBr4]2−
1 Both S-I and S-II are correct
2 Both S-I and S-II are incorrect
3 S-I is correct, and S-II are incorrect
4 S-I is incorrect,t and S-I is correct
Question 3: A rectangle is formed by lines x=0,y=0,x=3,y=4. A line perpendicular to 3x+4y+6=0 divides the rectangle into two equal parts, then the distance of the line from (−1,32) is
1) 2
2) 17/10
3) 6/5
4) 8/5
Question 4: Let $A=\{-2,-1,0,1,2,3,4\}$ and $R$ be a relation $R$, such that $R=\{(x, y):(2 x+y) \leq-2, x \in A, y \in A\}$.
Let I = number of elements in Rm == minimum number of elements to be added in R to make it reflexive =
n= minimum number of elements
be added in R to make it symmetric, then (1+m+n) is
1)10
2)17
3)11
4) 14
Question 5: Determine the correct energy for the ground stateor the energy transition. (symbols have usual meaning & n→m gives the transition)1) $\mathrm{H}(-6.8 \mathrm{eV})$
2) $\mathrm{Li}^{2+}(-13.6 \mathrm{eV})$
3) $\mathrm{He}_{2 \rightarrow 1}^{+}(40.8 \mathrm{eV})$
4) $\mathrm{Be}_2^{3+} 1(+13.6 \mathrm{eV})$
The JEE Mains 2026 Jan 23 Shift 2 (Evening shift) was moderate. You can also check the JEE Main 2026 January 23 Shift 1 Question Paper.
Question 1. Match List-I with List-II.
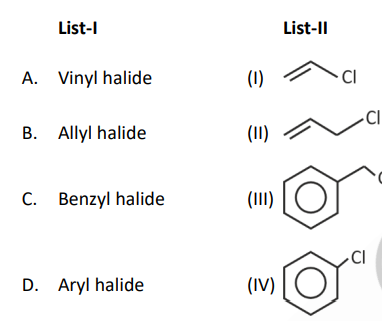
Select the correct option.
(1) A(II), B(I), C(III), D(IV)
(2) A(I), B(II), C(III), D(IV)
(3) A(I), B(II), C(IV), D(III)
(4) A(II), B(I), C(IV), D(III)
Answer (2)
Question 2. The correct order of stability of the following diazonium ions is
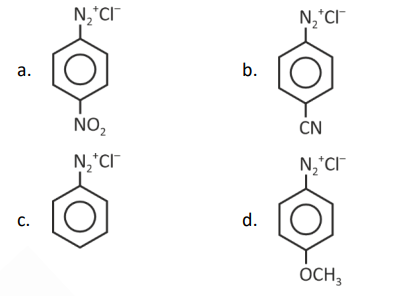
(1) a < b < c < d
(2) a < b < d < c
(3) c < d < b < a
(4) d < c < b < a
The stronger the electron-withdrawing group attached to the para position of −N2+in diazonium ion, the less is the stability and the more electrophilic it is.
Stability: (d)>(c)>(b)>(a)
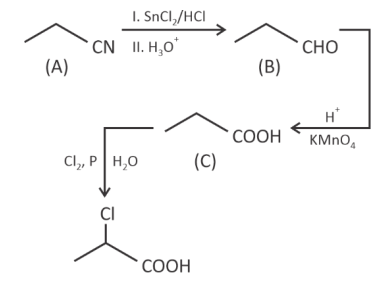
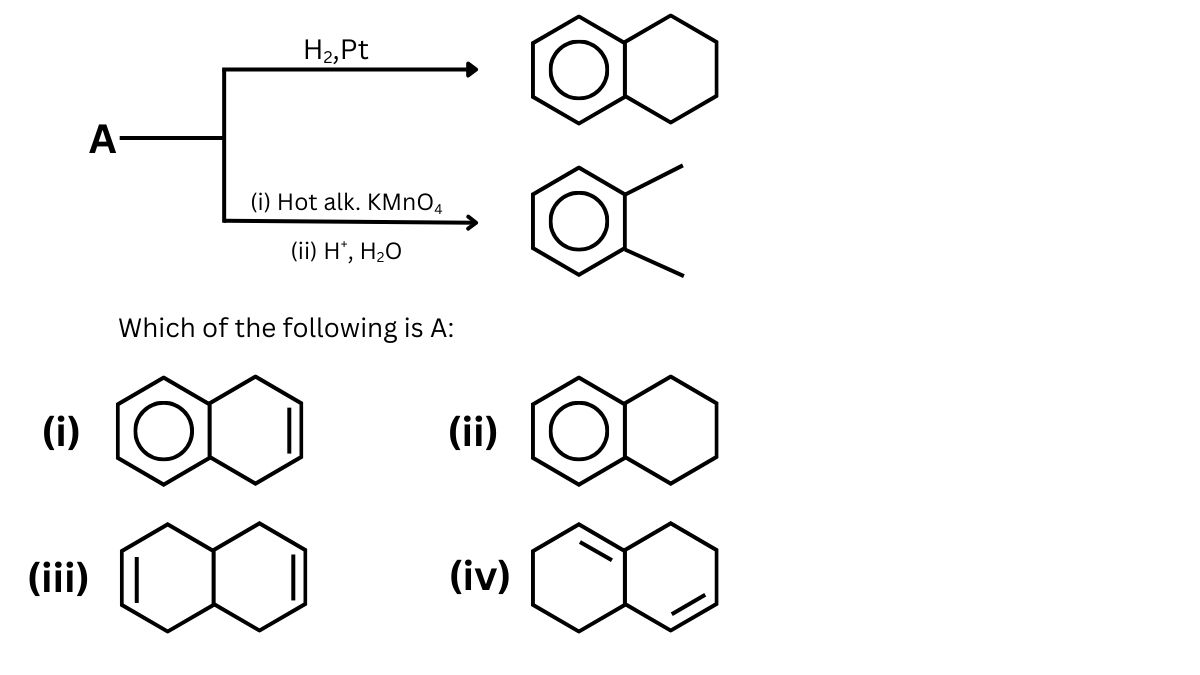
Question 3.
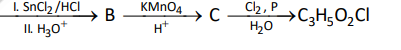
The final product has one chiral centre. Structure of A is
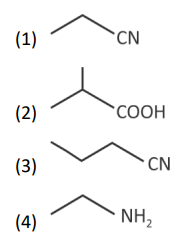
Answer (1)
Question 4. Which of the following compounds contains 3 unpaired electrons?
(1) $\mathrm{V}_2 \mathrm{O}_5$
(2) $\left[\mathrm{TiF}_6\right]^{3-}$
(3) $\left[\mathrm{CoF}_6\right]^{4-}$
(4) $\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{3-}$
Answer (3)
$\mathrm{V}_2 \mathrm{O}_5: 0$ unpaired electrons
$\left[\mathrm{TiF}_6\right]^{3-}: \mathrm{Ti}^{3+}:[\mathrm{Ar}] 4 s^0 3 d^1: 1$ unpaired $\mathrm{e}^{-}$
$\left[\mathrm{CoF}_6\right]^{4-}: \mathrm{Co}^{2+}:[\mathrm{Ar}] 4 s^0 3 d^7: 3$ unpaired $\mathrm{e}^{-}$
$\left[\mathrm{Fe}(\mathrm{CN})_6\right]^{3-}: \mathrm{Fe}^{3+}:[\mathrm{Ar}] 4 s^0 3 d^5: 1$ unpaired $\mathrm{e}^{-}$
Question 5. Consider the following molecules.
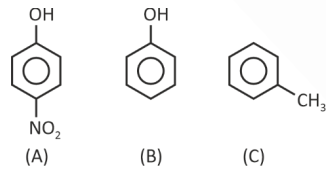
The correct order of dipole moment is
(1) A > B > C
(2) A > C > B
(3) B > A > C
(4) C > A > B
Answer (1)
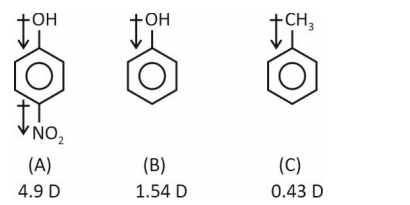
Dipole moment A > B > C
The JEE Mains 2026 Jan 22 evening shift was slightly tougher than the morning shift, with Physics being the toughest of all three. Maths was easy, chemistry was moderate, and overall, the paper was moderate. You can view the in-depth information by downloading the JEE Mains 2026 January 22 Shift 2 Question Paper analysis.
| Subject | Difficulty Level |
|---|---|
| Physics | Easy to Moderate |
| Chemistry | Moderate |
| Mathematics | Difficult |
| Overall | Moderate |
Some questions from Jan 22 Shift 2 are listed below
Question 1:The correct order of electron gain enthalpy (magnitude only) for group 16 elements is
1) Te > Se > S > O
2) S > Se > Te > 0
3) O > S > Se > Te
4) S > O > Se > Te
Question 2: 100 g 98 % by weight H 2 SO 4 is mixed with 100 g 49 % by weight H 2 SO 4 . Mole fraction of H 2 SO 4 solution is
1 0.9
2 0.1
3 0.67
4 0.33
Question 3: 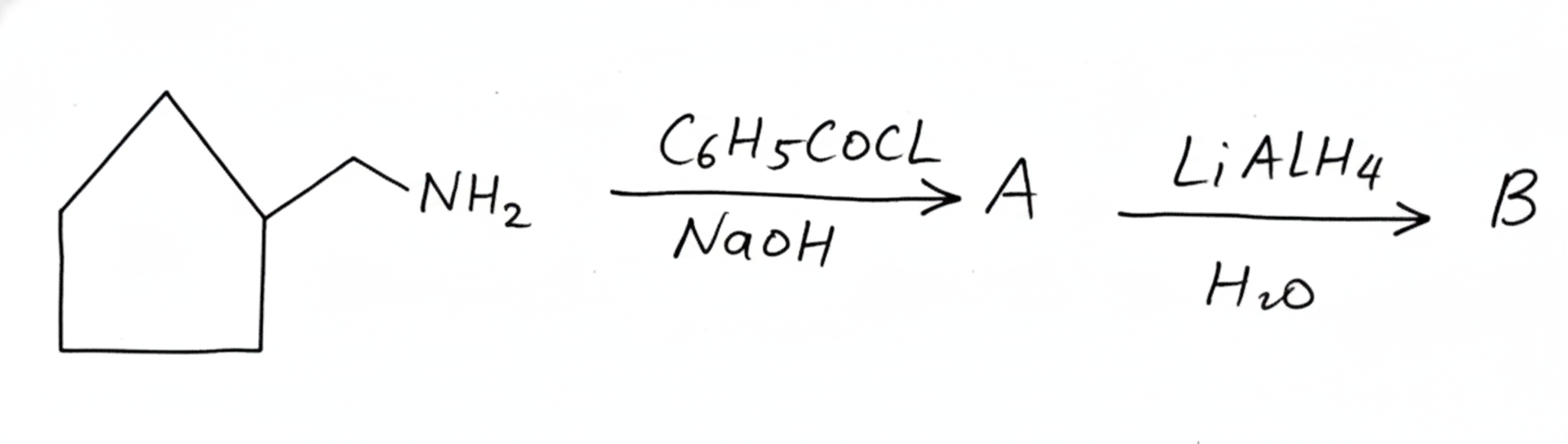
Question 4:
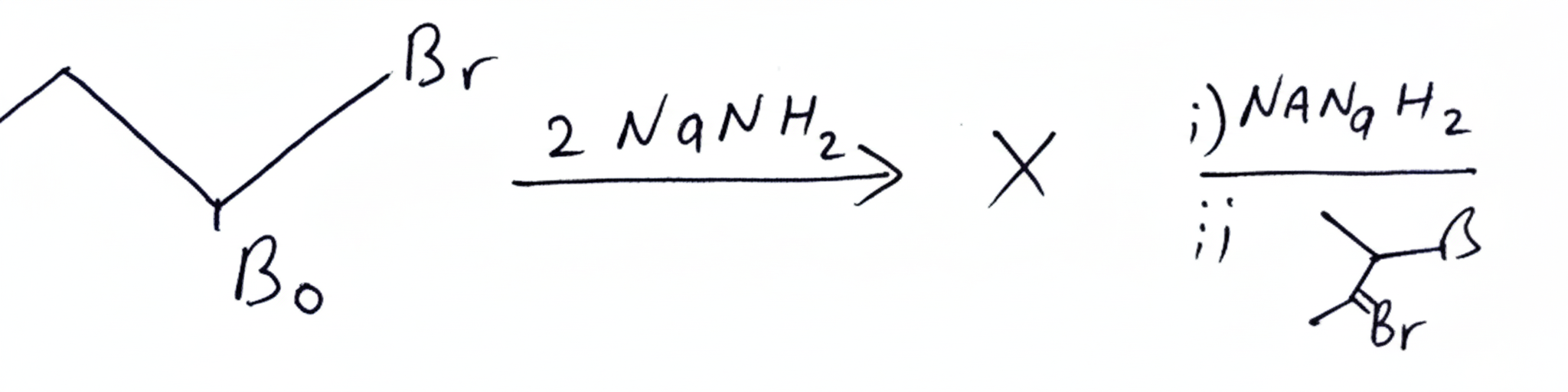
(1) isopropyl but-1-yne
(2) 2-Methyl hex-2-yne
(3) 5-Methyl Hex-2-yne
(4) 2-Methyl hex - 3-yne
JEE Main 2026 – January 22 Morning Shift was moderate to tough, with Physics being the most challenging, Mathematics lengthy and calculation-intensive, and Chemistry comparatively easier and more scoring. You can download the JEE Mains 2026 January 22 Question paper Shift 1also.
| Subject | Difficulty Level | Key Observations |
|---|---|---|
| Physics | Moderate to Difficult | Conceptual and time-consuming; tougher than Maths and Chemistry |
| Mathematics | Moderate | Lengthy calculations required strong time management |
| Chemistry | Easy to Moderate | More scoring; Organic-heavy with direct questions |
Questions are provided below
Question 1: Consider the given central metal ions of low spin complex and choose the correct increasing order of unpaired electrons $\mathrm{Mn}^{3+}, \mathrm{Cr}^{3+}, \mathrm{Fe}^{3+}, \mathrm{Co}^{3+}$
(1) $\mathrm{Co}^{3+}<\mathrm{Fe}^{3+}<\mathrm{Mn}^{3+}<\mathrm{Cr}^{3+}$
(2) $\mathrm{Co}^{3+}<\mathrm{Mn}^{3+}<\mathrm{Fe}^{3+}<\mathrm{Cr}^{3+}$
(3) $\mathrm{Cr}^{3+}<\mathrm{Mn}^{3+}<\mathrm{Cr}^{3+}<\mathrm{Fe}^{3+}$
(4) $\mathrm{Cr}^{3+}<\mathrm{Mn}^{3+}<\mathrm{Co}^{3+}<\mathrm{Fe}^{3+}$
Question 2:

Question 3: Which of the following is the correct order of the reactivity of the given nucleophiles when treated with CH3Br in methanol?
$\mathrm{F}^{-}, \mathrm{I}^{-}, \mathrm{C}_2 \mathrm{H}_5 \mathrm{O}, \mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}$
1) $\mathrm{I}^{-}>\mathrm{C}_2 \mathrm{H}_5 \mathrm{O}^{-}>\mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}>\mathrm{F}^{-}$
2) $\mathrm{I}^{-}>\mathrm{F}^{-}>\mathrm{C}_2 \mathrm{H}_5 \mathrm{O}^{-}>\mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}$
3) $\mathrm{I}^{-}>\mathrm{C}_2 \mathrm{H}_5 \mathrm{O}^{-}>\mathrm{F}^{-}>\mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}$
4) $\mathrm{C}_6 \mathrm{H}_5 \mathrm{O}^{-}>\mathrm{F}^{-}>\mathrm{I}^{-}>\mathrm{C}_2 \mathrm{H}_5 \mathrm{O}^{-}$
Question 4: For the reaction given below at 25∘C
$$
\mathrm{A}_2 \rightleftarrows 2 \mathrm{~A}
$$
Find $\ln \mathrm{K}_{\mathrm{p}}$
Given $\left(\Delta G_f^{\circ}\right)_A=-50.384 \mathrm{~kJ} / \mathrm{mol}$
$$
\left(\Delta G_f^{\circ}\right)_{A_2}=-100 \mathrm{~kJ} / \mathrm{mol}
$$
(1) 0.43
(2) 0.23
(3) 0.31
(4) 0.53
Question 5:
Statement I: Sucrose is dextrorotary, a nd upon hydrolysis, it becomes laevorotatory.
Statement-II Sucros,e on hydrolysis gives glucose and fructose such that the laevorotation of glucose is more than the dextrorotation of fructose.
1 Both Statement I and Statement II are correct
2 Both Statement I and Statement II are incorrect
3 Statement I is correct, Statement II is incorrect
4 Statement-II is correct, Statement-I is incorrect
You can get the details by downloading the JEE Main 2026 January 21 Question Paper Shift 2 with Solutions. The paper was easy to moderate. The level of difficulty matched that of the January 2025 sessions. Among the three subjects, Chemistry was the easiest, followed by Mathematics. Physics was the most scoring. Find more details below:
Subject | Overall Difficulty | Nature of Questions | Key Observations |
|---|---|---|---|
Physics | Easy to Moderate | Mostly formula-based, direct | Easier than the morning shift and the previous year; doable with proper revision |
Easy | Conceptual and statement-based | Easiest among all three subjects; revision-focused questions | |
Moderate to Hard | Lengthy and calculative | More time-consuming; required strong accuracy and time management |
Some memory-based questions are given below
Question 1: 1 g of an organic compound produces 1.49g of Mg2P2O7. Determine % of P
Question 2: Given below are two statements
Statement-I : The correct order for radius is $\mathrm{Al}>\mathrm{Mg}>\mathrm{Mg}^{2+}>\mathrm{A}^{\3+}$
Statement-II: Atomic size always depends on electronegativity.
In light of the above statements, choose the correct option.
1 Statement I and II are correct
2 Both Statement I and II are incorrect
3 Statement I is correct, bbut StatementII is incorrect
4 Statement I is incorrect, but Statement II is correct
Question 3: What will be the significant figure of the summation of 0.153,153.2 and 153.2?
1) 3
2) 4
3) 5
4) 6
Question 4: When 8.74 g $8.74 \mathrm{gMnO}_2$ is treated with HCl, then what will be the weight of $\mathrm{Cl}_2(\mathrm{~g})$ obtained? Molar mass of $\mathrm{MnO}_2=87.4 \mathrm{~g} / \mathrm{mol}$
17.1 g
217.1 g
314.2 g
43.55 g
Question 5: The rms speed of oxygen molecules at 47∘C is equal to that of hydrogen molecules kept at $\circ C\left(\frac{M(0)}{M(h)}=\frac{32}{2}\right)$
(A) -100
(B) -253
(C) -20
(D) -235ow:
This shift was slightly tougher in terms of difficulty than last year. Some questions will be given below soon. You can check the JEE Mains Jan 21 Shift 1 Question Paper and the analysis table below:
| Parameter | Shift 1 |
|---|---|
| Overall Difficulty Level | Moderate to Tough |
| Physics Difficulty | Moderate |
| Chemistry Difficulty | Tough |
| Mathematics Difficulty | Moderate (easier than last year) |
| Numerical-Based Questions | Moderate number (mainly in Physics & Maths) |
| Conceptual Questions | High (especially in Chemistry) |
Some memory-based questions are given below
Question 1:
Solution: Option (1) is correct
Question 2: In Sulphur estimation, 0.7 g of an organic compound gives 1 g of BaSO4 by the Carius method. What is the % of ‘S’ in the compound?
(1) 19.61
(2) 23.85
(3) 27.93
(4) 14.57
Answer: Correct answer is option (1)
Question 3: Which of the following is the correct order with respect to the property indicated?(1) CI $>$ F (Ionisation energy)
(2) $\mathrm{K}_2 \mathrm{O}>\mathrm{Na}_2 \mathrm{O}>\mathrm{Al}_2 \mathrm{O}_3$ (Basic nature)
(3) $\mathrm{K}>\mathrm{Na}>\mathrm{Al}>\mathrm{Mg}$ (Metallic character)
(4) None of these
Answer: Correct answer is option (2)
Question 4: In 'S' estimation 0,.7 g of an organic compound gives 1 g BaSO4. In the Carius method. What is the % of ' S ' in the compound?
1) 19.61
2) 23.85
3) 27.93
4) 14.57
Question 5: Consider the following reaction
$\mathrm{Ca}+2 \mathrm{HCl} \rightarrow \mathrm{CaCl}_2+\mathrm{H}_2$
We have 14 g of Ca react with an excess of HCl. Choose the incorrect option.
(1) Mass of CaCl2 produced is 38.85 g
(2) Mole of H2 produced is 0.35 mol
(3) Volume of H2 produced at STP is 7.84 L
(4) Mass of CaCl2 produced is 3.885 g
Answer: Correct answer is option (4)
Question 6: Given below are two statements.
Statement 1: Arginine and Tryptophan are essential amino acids.
Statement 1: Glycine does not have any chiral carbon.
In light of the above statements, which is the correct option?
Both statements I and II are correct
2 Both statements I and II are incorrect
3 Statement I is correct, and Statement II is incorrect
4 Statement-I is incorrect and Statement-II is correct:
The previous year's question paper, i.e., JEE Mains 2025 question paper, must be solved by students before appearing for the JEE Mains 2026 paper. It is an important study material for students, which will help them understand how their preparation is coming along. You can view the JEE Mains 2025 question paper below:
In this section,n we will see the JEE Mains previous years' question paper. This is to familiarise yourself with the type of questions to expect in your exam. There have been changes in the syllabus, so you must only solve questions that are included in the JEE Mains 2026 syllabus. You can download the JEE Mains 2026 paper from the link below:
If you are studying subject-wise and want to download the JEE 2026 Mains model question paper with detailed answers per subject, we have given it below:
You can also practice the JEE Mains 2026 sample paper:
Let us understand the JEE Mains exam pattern and also go through JEE Mains 2026 model questions:
Note: Incorrect answer will have negative marking of 1 from MCQs, but there is no negative marking in case of incorrect answers in numerical questions.
Subject | Section | Question Type | No. of Questions | Marks |
Mathematics | Section A | MCQs | 20 | 80 |
Section B | Numerical Value | 5 | 20 | |
Physics | Section A | MCQs | 20 | 80 |
Section B | Numerical Value | 5 | 20 | |
Chemistry | Section A | MCQs | 20 | 80 |
Section B | Numerical Value | 5 | 20 | |
Total | 75 | 300 Marks |
Frequently Asked Questions (FAQs)
Yes, the JEE Main 2026 exam is going to be prepared by the NTA based on the same syllabus, unless changed, using the textbooks of NCERT.
You will get a total of 75 questions (25 each in the three subjects).
It will be carried out in two sessions- probably in January and April next year, 2026.
On this page, you can get PDF files of past years question papers with detailed solutions of last 10 years paper to refer easily. But for 2026, the JEE Main 2026 question paper with answers will be officially released by NTA a few days after the exam.
On Question asked by student community
Hello Dear Student,
Yes, with an SC category rank of 31,370, you have a very strong chance of getting admission to IIITDM Kancheepuram. While highly demanding branches like Computer Science and Engineering may close earlier (around 3,400 - 92,000 depending on the round), your rank is highly competitive for other
Hello Dear Student,
With a JEE Main CRL of 1,22,306 and an OBC rank of 40,694, securing a seat in top-tier NITs or IIITs for core branches will be highly challenging. However, using your Gujarat Home State Quota, your best options include GFTIs, newer IIITs, state-funded universities, and reputable private
Hello,
With 982/1000 in IPE (98.2%) and 89 percentile in JEE Main , you have a good chance of getting a seat at SASTRA University , especially through the admission process that considers both Class 12 marks and JEE Main scores. Your excellent board marks will work strongly in your
Hello,
With a CRL rank of 1.57 lakh , OBC rank of 54,000 , and Haryana domicile , getting CSE at YMCA Faridabad is unlikely based on previous cutoff trends.
However, you may still have a chance in:
Electronics & Computer Engineering (depending on cutoff movement)
Hello Dear Student,
With an OBC-NCL category rank of 25,995 in JEE Main , your chances at the most sought-after branches in top IIITs are limited, but you still have several realistic options through JoSAA and especially CSAB Special Rounds .
You may have chances in:
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Top Placements: 50 LPA in Google | 46.38 LPA in Amazon | 45 LPA in Adobe | 50 LPA in Microsoft | 44.14 in Amazon
40 LPA Highest Package | Up to 100% Scholarship worth 24 Crore via GUTS exam
100+ Recruiters | 100% Placement Assistance | Scholarship Available for Meritorious Students
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score