Amity University Noida-B.Tech Admissions 2026
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
The Joint Entrance Examination (JEE) Main is conducted by the National Testing Agency (NTA), and every year, lakhs of students appear for the exam. It is the entrance test – or the preliminary selection round – for the top engineering colleges in the country including the Indian Institutes of Technology (IIT), National Institutes of Technology (NIT), Indian Institutes of Information Technology (IIIT) and many more government institutions.

Clearly, JEE Main is critical. However, JEE Main 2021 had questions which were conceptually or factually ambiguous and some of them were simply incorrect, going by the JEE Main answer key. Last year, the exam was conducted in four phases – February, March, July and August – across 26 Shifts. The result was declared in September.
Here, Careers360 looks into the questions asked in the July and August shifts, and highlights some of the errors in the JEE Main previous year question paper’s Chemistry section. The necessary changes required for these questions were absent from the NTA JEE Main answer keys, which could have affected the scorecards of many candidates in the JEE Main result.
These questions, along with their respective problems, are listed out below:
Question ID : 86435117255
JEE Main Answer Key: 172

The issue:
The unit in which Kp is to be reported has not been specified in the question.
So, a student solving for the unit as (Pa)-1 will get an answer of 0.172 which would be rounded off to 0 while the given answer will be in units of (kPa)-1

____________________________________________________________________________
Question ID : 86435117528
JEE Main Answer Key: 1

The issue:
There is a conceptual error in the question. A negatively-charged sol cannot be precipitated by a negatively-charged ion.
Also, the coagulating value is defined as the minimum concentration of an electrolyte in millimoles per litre required to cause precipitation of a sol in two hours.
So, there arises another problem: How do you extrapolate data for two hours from the one-hour data?
____________________________________________________________________________
Question ID : 86435117785
JEE Main Answer Key: 86435159952 (Option 2)
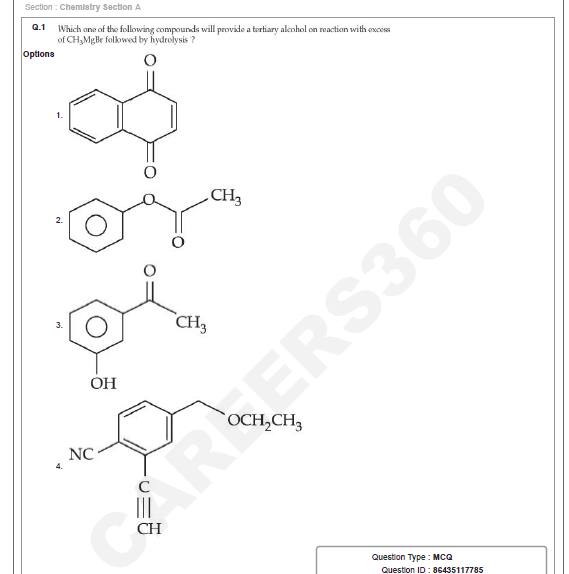
Question and Option IDs

The issue:
Multiple options given in the question (Options 1,2 and 3) are capable of forming tertiary alcohol on reaction with excess of Grignard reagent (CH3MgBr) followed by hydrolysis.
Option 1: Alpha- Beta unsaturated carbonyl compounds show direct addition to the carbonyl group with Grignard reagent. Hence, tertiary alcohols will be obtained from the carbonyl groups
Option 3: Grignard reagent can add to a ketone, forming a tertiary alcohol.
____________________________________________________________________________
Question ID : 86435118234
JEE Main Answer Key: 86435161299 (Option 1)
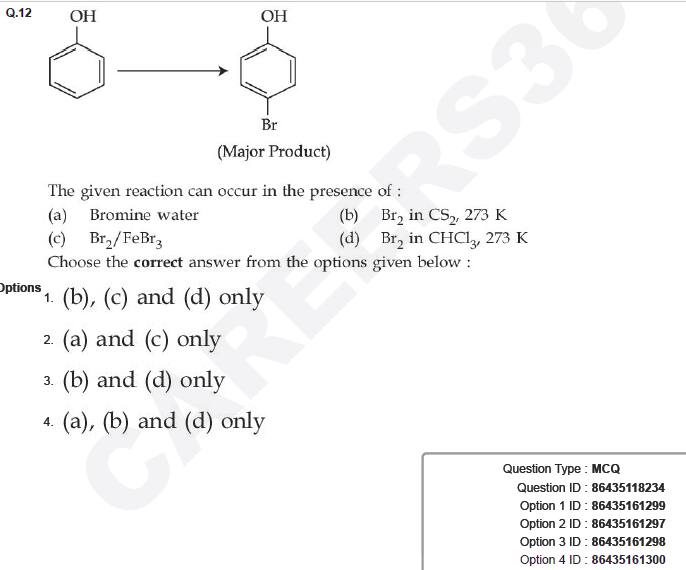
The issue:
Lewis acids are not used with Phenol because Phenol itself forms a complex with the Lewis acid, due to which the ring becomes deactivated towards Electrophilic Substitution.
In fact, this reaction of Phenol with a Lewis acid FeCl3 is actually used to detect the presence of Phenol in solution. Phenol forms a complex with neutral FeCl3 and produces a violet colour.
Correct answer should be 86435161298 [Option (3)]
____________________________________________________________________________
Question ID : 86435118225
JEE Main Answer Key: 86435161264 (Option 3)

The issue:
Be is not an alkaline earth metal as its oxide and hydroxide are Amphoteric in nature.
Here is an excerpt from NCERT textbook’s chapter on the s Block elements confirming it.

Hence, Statement I is also correct in the given question.
Correct answer should be 86435161261 [Option (2)]
____________________________________________________________________________
Question ID : 86435118870
JEE Main Answer Key: 5

The issue:
Data given is insufficient to solve the problem.
Molality (m) can be calculated from the density of solution only when the Molarity (M) is also given in the question.
In the question, Molarity has not been provided and thus the question cannot be solved.
____________________________________________________________________________
Question ID : 86435119236
JEE Main Answer Key: 300

The issue :
The answer in the JEE Main final answer key was wrong. The answer should have been 600.

____________________________________________________________________________
Get expert advice on college selection, admission chances, and career path in a personalized counselling session.
Question ID : 86435120226
JEE Main Answer Key: 50

The issue :
The answer given in the answer key is wrong. The answer should be 0.

The answer is 0.02 which would be rounded off to 0.
For the answer to be 50, the value of (Kc)-1 had to be asked.
____________________________________________________________________________
Question ID : 86435120586
JEE Main Answer Key: 4

The issue :
The answer given is 4 because the inner 4f electrons have not been counted.
The question does not say that we need to consider only the 5f electrons
____________________________________________________________________________
Question ID : 86435121290
JEE Main Answer Key: 86435170463 (Option 4)
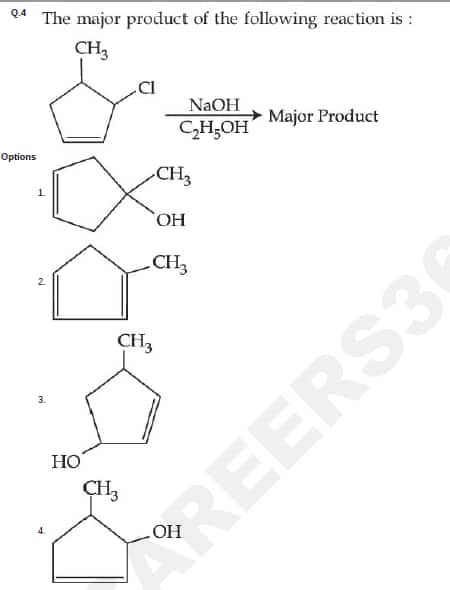
The issue :
Alcoholic NaOH causes dehydrohalogenation (elimination) and not substitution.
The answer should be 86435170462 (Option 2)
____________________________________________________________________________
Question ID : 86435121287
JEE Main Answer Key: 86435170452 (Option 2)

The issue :
None of the options is correct.
All the given options contain permanganate ion (MnO4-) which contains Mn in its highest oxidation state (+7) and hence, it cannot show disproportionation reaction
Question should have been awarded as bonus
____________________________________________________________________________
Question ID : 86435121562
JEE Main Answer Key: 86435171281 (Option 4)
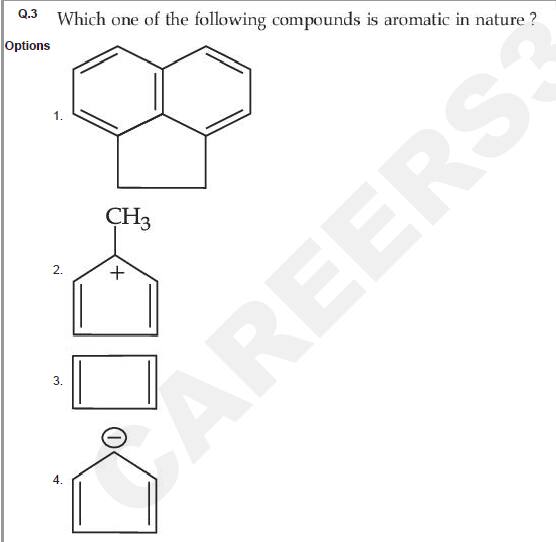
The issue :
Options 1 and 4 can both be correct but only Option 4 has been given as the correct answer.
Acenaphthene (compound in Option 1) is also Aromatic in nature.
____________________________________________________________________________
Apart from the above-mentioned questions, there were also some in the integer section that had very lengthy mathematical calculations and whose answers had to be rounded off to the nearest integers.
These questions are usually solved by approximations and it is very difficult for students to arrive at the correct integer answer. Previously, these questions had a correct range for the answer, which gave students some leeway to make appropriate approximations in calculations.
Now, with negative marking in the numerical response questions, this becomes an even bigger problem. Considering the high-stakes nature of the JEE Mains, even a few such errors can have significant ramifications for applicants and be deeply demoralising.
On Question asked by student community
Hello Dear Student,
Yes, with an SC category rank of 31,370, you have a very strong chance of getting admission to IIITDM Kancheepuram. While highly demanding branches like Computer Science and Engineering may close earlier (around 3,400 - 92,000 depending on the round), your rank is highly competitive for other
Hello,
With 982/1000 in IPE (98.2%) and 89 percentile in JEE Main , you have a good chance of getting a seat at SASTRA University , especially through the admission process that considers both Class 12 marks and JEE Main scores. Your excellent board marks will work strongly in your
Hello,
With a CRL rank of 1.57 lakh , OBC rank of 54,000 , and Haryana domicile , getting CSE at YMCA Faridabad is unlikely based on previous cutoff trends.
However, you may still have a chance in:
Electronics & Computer Engineering (depending on cutoff movement)
Hello Dear Student,
With an OBC-NCL category rank of 25,995 in JEE Main , your chances at the most sought-after branches in top IIITs are limited, but you still have several realistic options through JoSAA and especially CSAB Special Rounds .
You may have chances in:
Hello,
With an OBC-NCL rank of 25,995 and MP domicile , getting CSE in top IIITs may be difficult, but you still have chances in some newer IIITs and GFTIs, especially in later JoSAA and CSAB rounds.
You can consider:
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
Last Date to Apply: 15th June | Ranked #43 among Engineering colleges in India by NIRF | Highest Package 1.3 CR , 100% Placements
Top Placements: 50 LPA in Google | 46.38 LPA in Amazon | 45 LPA in Adobe | 50 LPA in Microsoft | 44.14 in Amazon
40 LPA Highest Package | Up to 100% Scholarship worth 24 Crore via GUTS exam
100+ Recruiters | 100% Placement Assistance | Scholarship Available for Meritorious Students
Mark presence in the Modern Architectural field with Bachelor of Architecture | Highest CTC : 70 LPA | Accepts NATA Score