Amity University Noida-B.Tech Admissions 2026
Among top 100 Universities Globally in the Times Higher Education (THE) Interdisciplinary Science Rankings 2026
16 Questions around this concept.
In the following structures, which one is having staggered conformation with maximum dihedral angle?
Arrange the following conformational isomers of n-butane in order of their increasing potential energy :
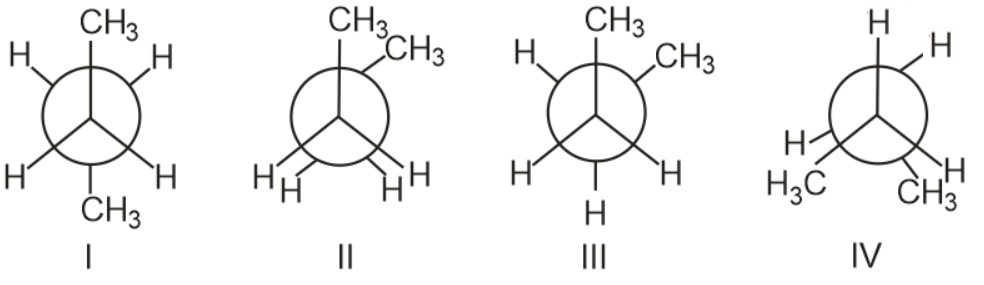
Which one of the following conformer of butane is most stable ?
JEE Main 2026: College Predictor | Official Question Papers
New: Apply to Multiple B.Tech Colleges Through Free 1:1 Counselling
Comprehensive Guide: IIT's | NIT's | IIIT's | Foreign Universities in India
In the given conformation $C_2$ is rotated about $C_2-C_3$ bond anticlockwise by an angle of $120^{\circ}$ then the formation obtained is

In the following the most stable conformation of n-butane is:
The incorrect statement regarding conformations of ethane is :
Newman projection P, Q, R and S are shown below. Which one of the following represent identical molecule?
The most stable conformation for n-butane is
The most stable conformation of 2,3 -dibromobutane is
Which one of the following conformations of cyclohexane is chiral?
Conformations
Alkanes contain carbon-carbon sigma (σ) bonds. Electron distribution of the sigma molecular orbital is symmetrical around the internuclear axis of the C–C bond which is not disturbed due to rotation about its axis. This permits free rotation about C–C single bond. This rotation results into different spatial arrangements of atoms in space which can change into one another. Such spatial arrangements of atoms which can be converted into one another by rotation around a C-C single bond are called conformations or conformers or rotamers. Alkanes can thus have infinite number of conformations by rotation around C-C single bonds. However, it may be remembered that rotation around a C-C single bond is not completely free. It is hindered by a small energy barrier of 1-20 kJ mol–1 due to weak repulsive interaction between the adjacent bonds. Such a type of repulsive interaction is called torsional strain.
Sawhorse projections
In this projection, the molecule is viewed along the molecular axis. It is then projected on paper by drawing the central C–C bond as a somewhat longer straight line. Upper end of the line is slightly tilted towards right or left hand side. The front carbon is shown at the lower end of the line, whereas the rear carbon is shown at the upper end. Each carbon has three lines attached to it corresponding to three hydrogen atoms. The lines are inclined at an angle of 120° to each other. Sawhorse projections of eclipsed and staggered conformations of ethane are shown in the figure.

Newman projections
In this projection, the molecule is viewed at the C–C bond head on. The carbon atom nearer to the eye is represented by a point. Three hydrogen atoms attached to the front carbon atom are shown by three lines drawn at an angle of 120° to each other. The rear carbon atom is represented by a circle and the three hydrogen atoms are shown attached to it by the shorter lines drawn at an angle of 120° to each other. The Newman’s projections for ethane are shown in the figure.

"Stay in the loop. Receive exam news, study resources, and expert advice!"